Chapter 16: Q52P (page 784)
Question: Draw the structure for each of the following:
a.
b.
c.
d.
Short Answer
ANSWER
a
Learning Materials
Features
Discover
Chapter 16: Q52P (page 784)
Question: Draw the structure for each of the following:
a.
b.
c.
d.
ANSWER
a
All the tools & learning materials you need for study success - in one app.
Get started for free
Hydration of an aldehyde is also catalyzed by hydroxide ion. Propose a mechanism for the reaction.
Question:a.What would have been the product of the preceeding reaction with LiAlH4 if the keto group had not been protected?
b. What reagent could you use to reduce only the keto group?
How would you make the following compounds from N-benzylbenzamide? a. dibenzylamine b. benzoic acid c. benzyl alcohol
When a cyclic ketone reacts with diazomethane, the next larger cyclic ketone is formed. This is called a ring-expansion reaction. Draw a mechanism for the following ring-expansion reaction.
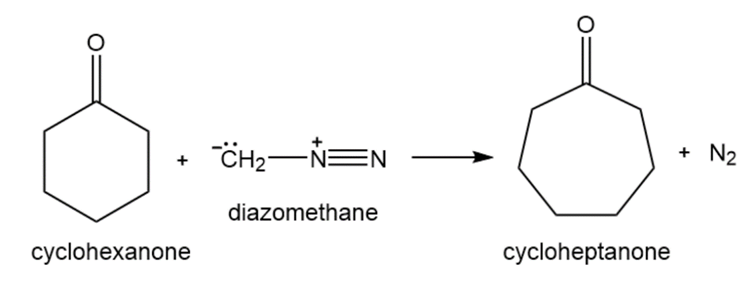
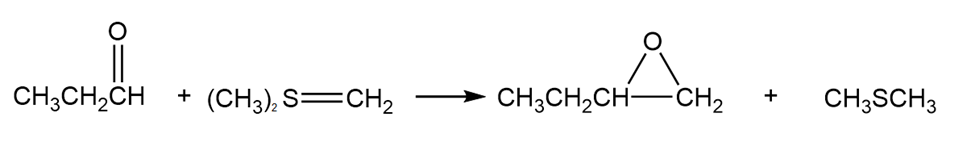
Unlike a phosphonium ylide that reacts with an aldehyde or a ketone to form an alkene, a sulfonium ylide reacts with an aldehyde or a ketone to form an epoxide. Explain why one ylide forms an alkene, whereas the other forms an epoxide.
What do you think about this solution?
We value your feedback to improve our textbook solutions.