Chapter 16: Q91P (page 798)
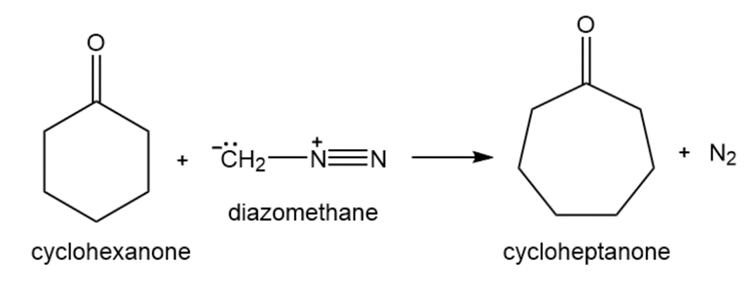
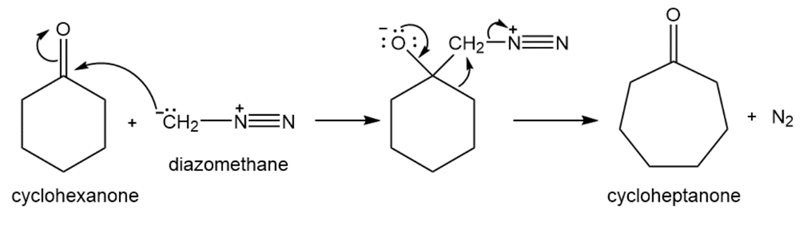
When a cyclic ketone reacts with diazomethane, the next larger cyclic ketone is formed. This is called a ring-expansion reaction. Draw a mechanism for the following ring-expansion reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 16: Q91P (page 798)
When a cyclic ketone reacts with diazomethane, the next larger cyclic ketone is formed. This is called a ring-expansion reaction. Draw a mechanism for the following ring-expansion reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
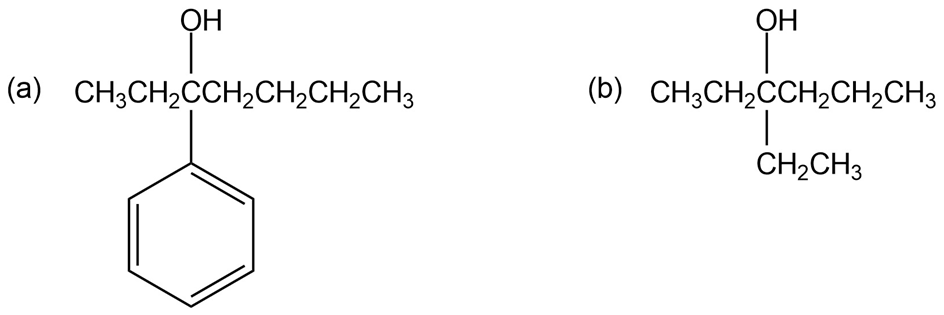
List three different sets of reagents (each set consisting of a carbonyl compound and a Grignard reagent) that could be used to prepare each of the following tertiary alcohols
Question:Can a cyanohydrin be prepared by treating a ketone with sodium cyanide?
Show how each of the following compounds can be prepared, using the given starting material:
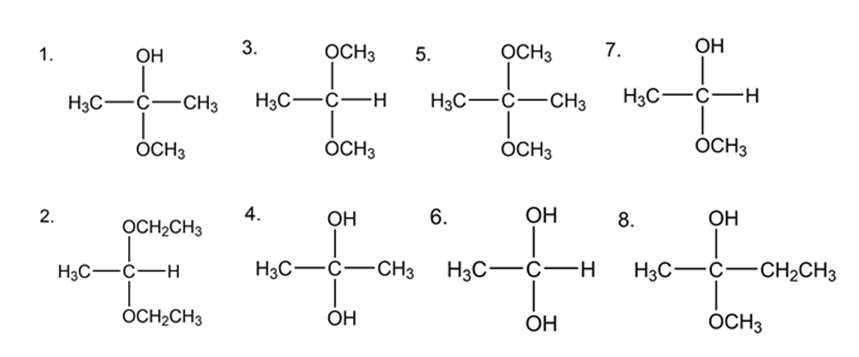
Which of the following are
a. hemiacetals?
b. acetals?
c. hydrates?
What are the products of the following reactions?
a.
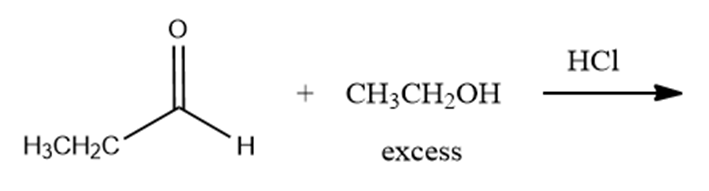
b.
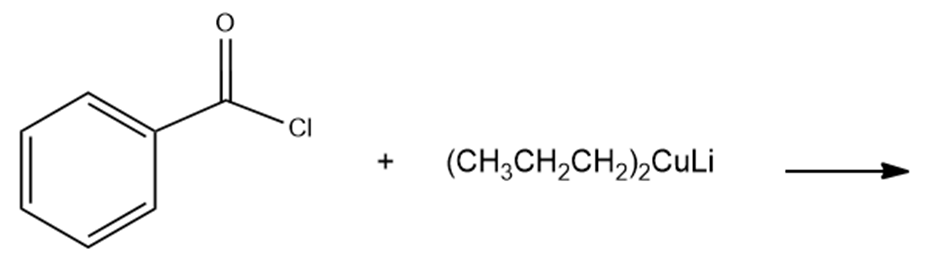
c.
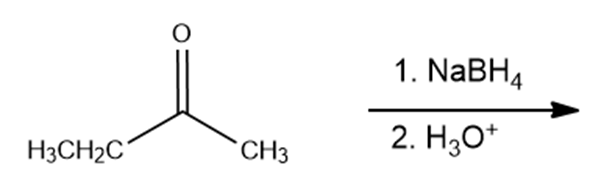
d.
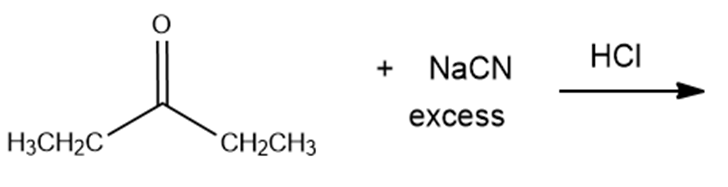
e.
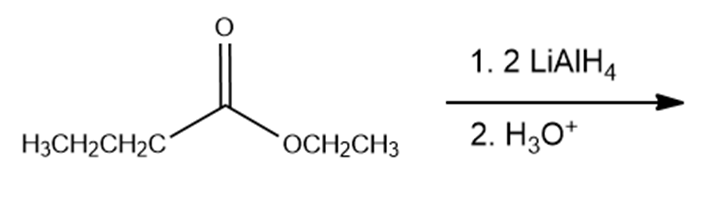
f.
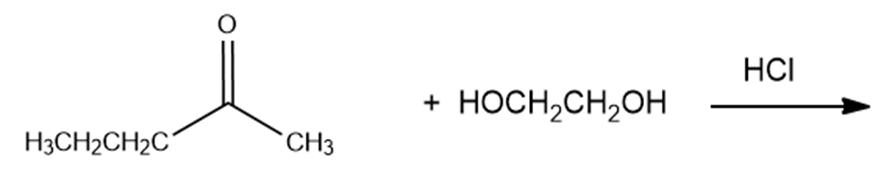
g.
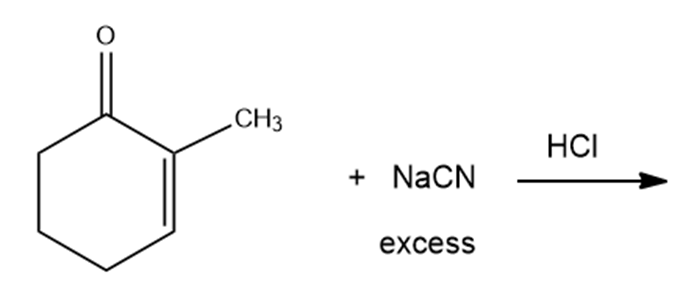
h.
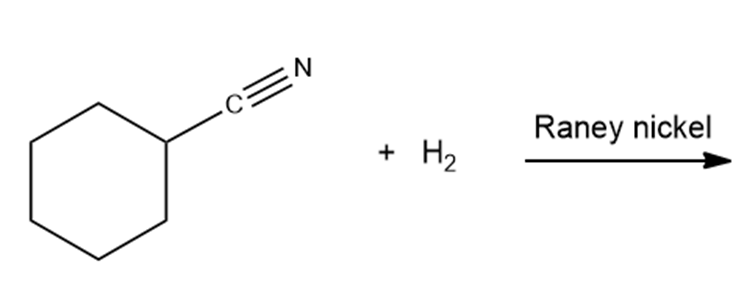
What do you think about this solution?
We value your feedback to improve our textbook solutions.