Chapter 16: Q16P (page 751)
Question:Can a cyanohydrin be prepared by treating a ketone with sodium cyanide?
Short Answer
Answer
A cyanohydrin can’t be prepared by treating a ketone with sodium cyanide.
Learning Materials
Features
Discover
Chapter 16: Q16P (page 751)
Question:Can a cyanohydrin be prepared by treating a ketone with sodium cyanide?
Answer
A cyanohydrin can’t be prepared by treating a ketone with sodium cyanide.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Thiols can be prepared from the reaction of thiourea with an alkyl halide, followed by hydroxide-ion-promoted hydrolysis.
a.Propose a mechanism for the reaction.
b.What thiol will be formed if the alkyl halide employed is pentyl bromide?
Question: How many signals would the product of the following reaction show in
a.its 1H NMR spectrum?
b.its 13C NMR spectrum?
Rank the following compounds from most reactive to least reactive towards nucleophilic addition.
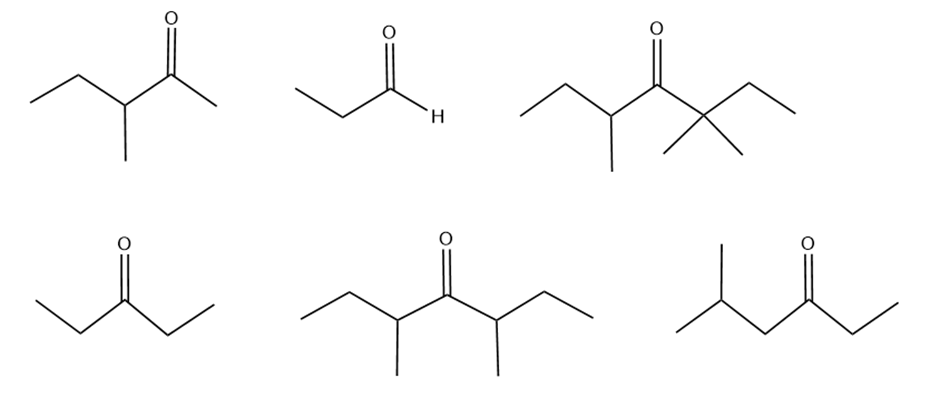
Explain why an acetal can be isolated but most hydrates cannot be isolated.
A compound gives the following IR spectrum. Upon reaction with sodium borohydride followed by acidification, it forms the product with the NMR spectrum shown below. Identify the starting material and the product.
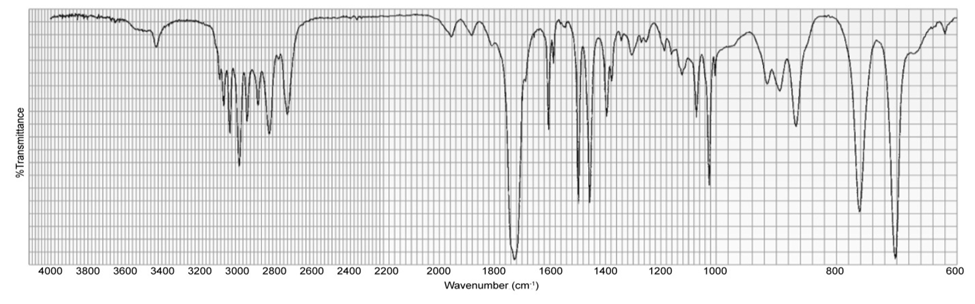
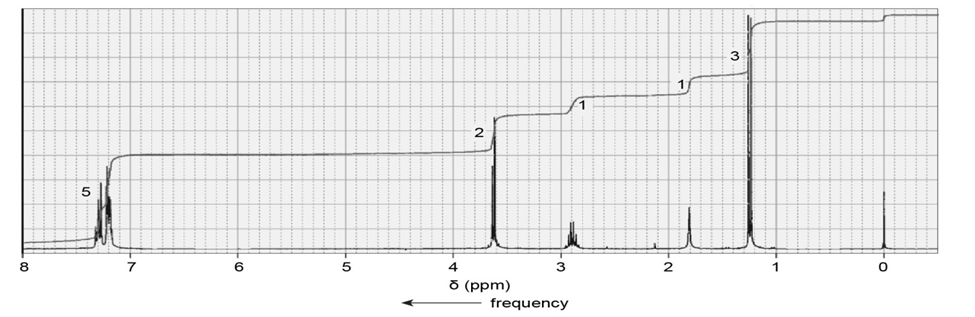
What do you think about this solution?
We value your feedback to improve our textbook solutions.