Chapter 16: Q39P (page 770)
Which of the following are
a. hemiacetals?
b. acetals?
c. hydrates?
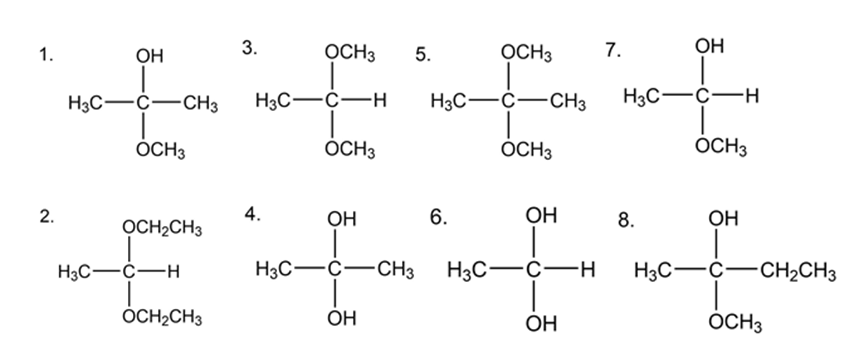
Short Answer
a. Hemiacetals- Compounds 1, 7, 8
b. Acetals- Compounds 2, 3, 5
c. Hydrates- Compounds 4, 6
Learning Materials
Features
Discover
Chapter 16: Q39P (page 770)
Which of the following are
a. hemiacetals?
b. acetals?
c. hydrates?

a. Hemiacetals- Compounds 1, 7, 8
b. Acetals- Compounds 2, 3, 5
c. Hydrates- Compounds 4, 6
All the tools & learning materials you need for study success - in one app.
Get started for free
a. Would you expect hemiacetals to be stable in basic solutions? Explain your answer.
b. Acetal formation must be catalyzed by an acid. Explain why it cannot be catalyzed by
c. Can the rate of hydrate formation be increased by hydroxide ion as well as by acid? Explain.
Question: Propose a mechanism for each of the following reactions:
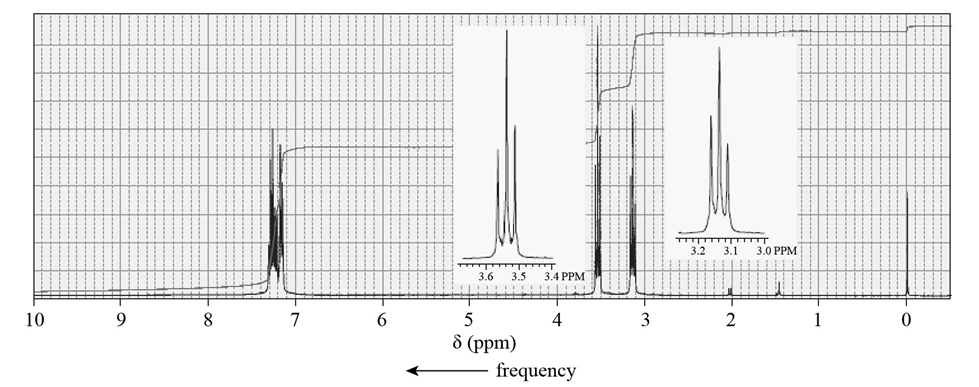
Shown below is the 1H NMR spectrum of the alkyl bromide used to make the phosphonium ylide that reacts with a ketone in a Wittig reaction to form acompound with molecular formula C11H14. What product is obtained from the Wittig reaction?
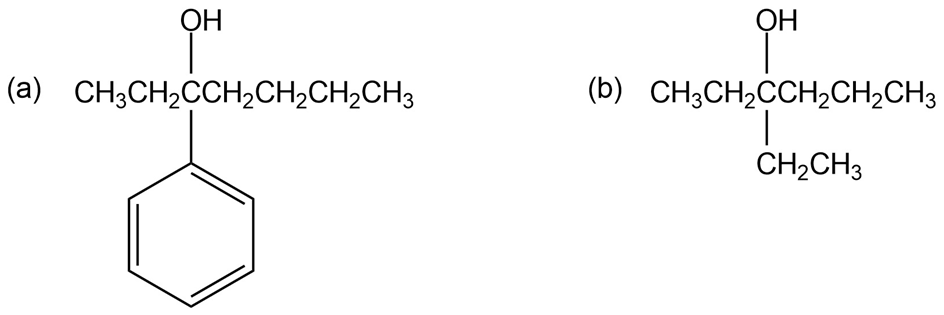
List three different sets of reagents (each set consisting of a carbonyl compound and a Grignard reagent) that could be used to prepare each of the following tertiary alcohols
Question: What are the products of the following reactions? (A trace amount of acid is present in each case.)
a. cyclopentanone + ethylamine
b. cyclopentanone + diethylamine
c. acetophenone + hexylamine
d. acetophenone + cyclohexylamine
What do you think about this solution?
We value your feedback to improve our textbook solutions.