Chapter 16: Q36P (page 767)
Hydration of an aldehyde is also catalyzed by hydroxide ion. Propose a mechanism for the reaction.
Short Answer
Mechanism of base catalyzed hydrolysis
Learning Materials
Features
Discover
Chapter 16: Q36P (page 767)
Hydration of an aldehyde is also catalyzed by hydroxide ion. Propose a mechanism for the reaction.
Mechanism of base catalyzed hydrolysis
All the tools & learning materials you need for study success - in one app.
Get started for free
a. In an aqueous solution, d-glucose exists in equilibrium with two six-membered ring compounds. Draw the structures of these compounds.
b. Which of the six-membered ring compounds will be the major product?
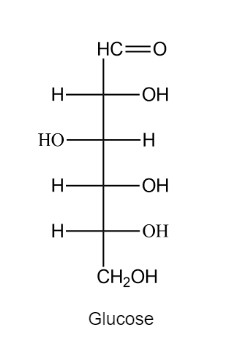
In the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces sleep when it is administered to animals in large doses, paraldehyde is used as a sedative or hypnotic. Propose a mechanism for the formation of paraldehyde.

Which of the following compounds does not form an alcohol when it reacts with excess Grignard reagent?
A.
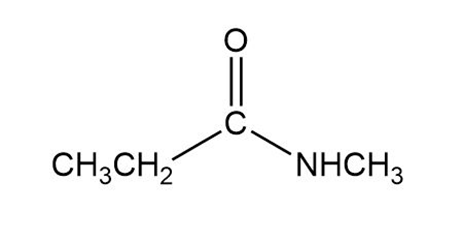
B.

C.

Shown below is the 1H NMR spectrum of the alkyl bromide used to make the phosphonium ylide that reacts with a ketone in a Wittig reaction to form acompound with molecular formula C11H14. What product is obtained from the Wittig reaction?
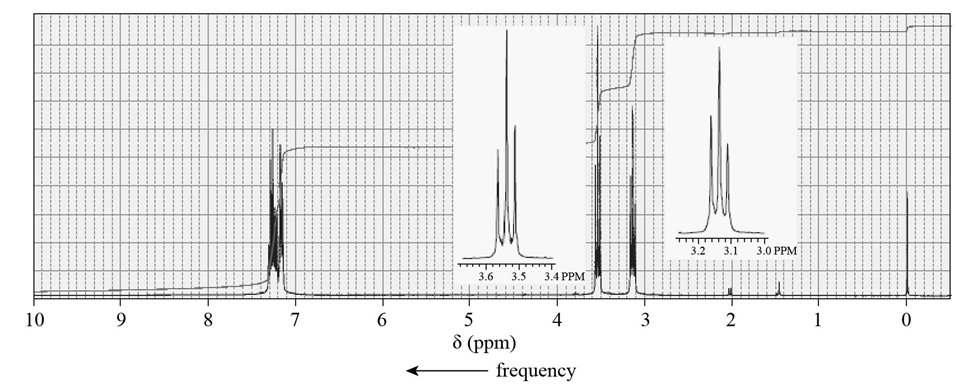
Question:a. In a six-step synthesis, what is the yield of the target molecule if each of the reactions employed gives an 80% yield?
b. What would the yield be if two more steps (each with an 80% yield) were added to the synthesis?
What do you think about this solution?
We value your feedback to improve our textbook solutions.