Chapter 16: Q37P (page 768)
Which ketone forms the most hydrate in an aqueous solution?
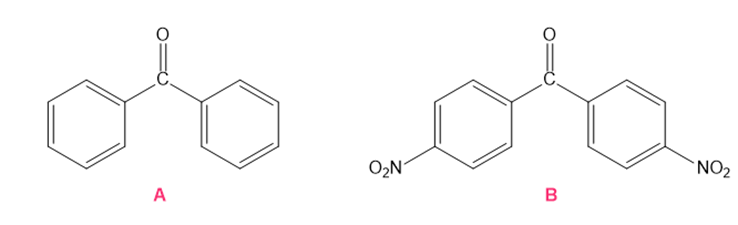
Short Answer
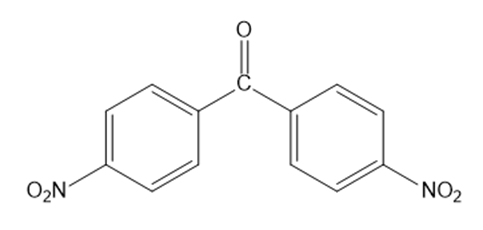
The ketone that forms the most hydrate
Learning Materials
Features
Discover
Chapter 16: Q37P (page 768)
Which ketone forms the most hydrate in an aqueous solution?


The ketone that forms the most hydrate
All the tools & learning materials you need for study success - in one app.
Get started for free
How would you make the following compounds from N-benzylbenzamide? a. dibenzylamine b. benzoic acid c. benzyl alcohol
In the mechanism of cyanohydrin formation, why is the only acid that protonates the alkoxide ion instead of ?
Shown below is the 1H NMR spectrum of the alkyl bromide used to make the phosphonium ylide that reacts with a ketone in a Wittig reaction to form acompound with molecular formula C11H14. What product is obtained from the Wittig reaction?

What is the product of each of the following reactions?
What alcohols are obtained from the Reduction of the following compounds with sodium borohydride?
a. 2-methylpropanal b. cyclohexanone c. 4-tert-butylcyclohexanone d. acetophenone
What do you think about this solution?
We value your feedback to improve our textbook solutions.