Chapter 16: Q2P (page 742)
Why are numbers not used to designate the position of the functional group in propanone and butanedione?
Short Answer
If the position of carbonyl group changes, then it would not be considered as a ketone.
Learning Materials
Features
Discover
Chapter 16: Q2P (page 742)
Why are numbers not used to designate the position of the functional group in propanone and butanedione?
If the position of carbonyl group changes, then it would not be considered as a ketone.
All the tools & learning materials you need for study success - in one app.
Get started for free
The pKa of protonated acetone is about -7.5, and the pKa of protonated hydroxylamine is 6.0.
a. In a reaction with hydroxylamine at pH 4.5 (Figure 16.2), what fraction of acetone is present in its acidic, protonated form? (Hint: See Section 2.10.)
b. In a reaction with hydroxylamine at pH 1.5, what fraction of acetone is present in its acidic, protonated form?
c. In a reaction with acetone at pH 1.5 (Figure 16.2), what fraction of hydroxylamine is present in its reactive basic form?
Question: a. Show the reagents required to form the primary alcohol in each of the following reactions.b. Which of the reactions cannot be used for the synthesis of isobutyl alcohol?
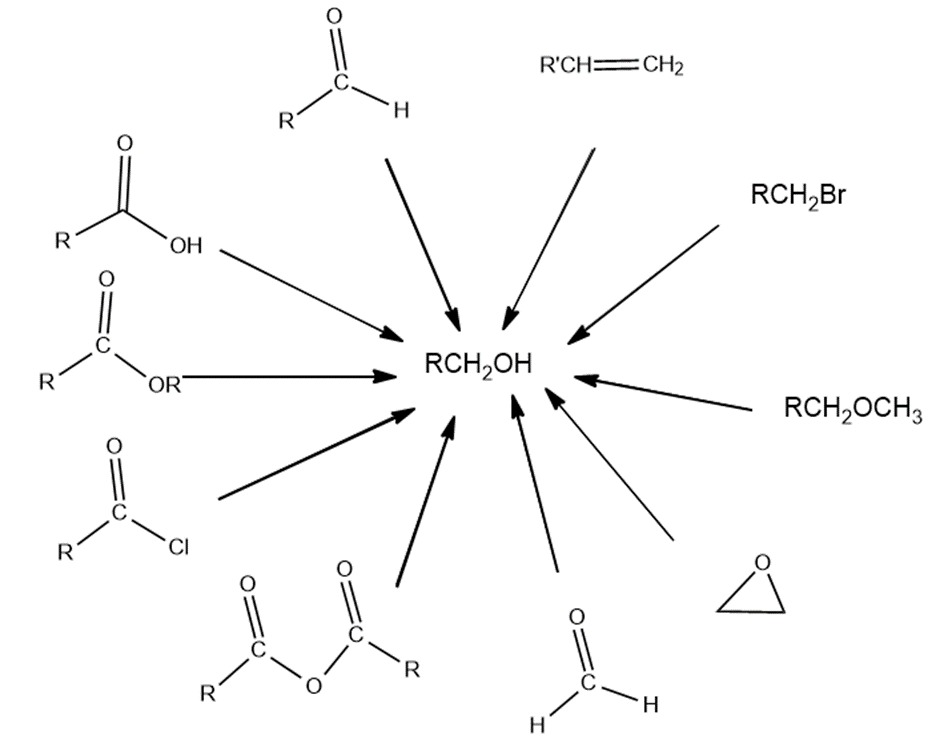
Draw the structure for each of the following:
a.
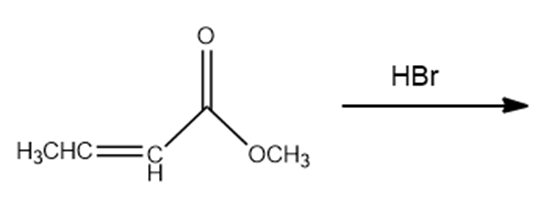
b.
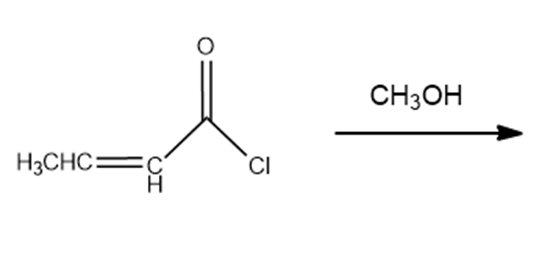
c.
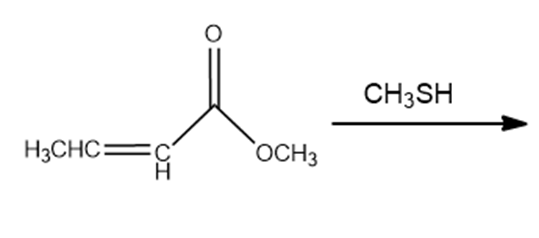
d.
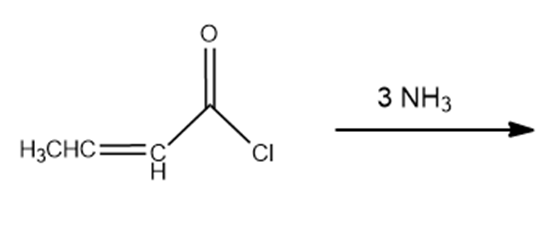
What is the product of the reaction of an ester with excess acetylide ion followed by the addition of pyridinium chloride?
What is the product of each of the following reactions?
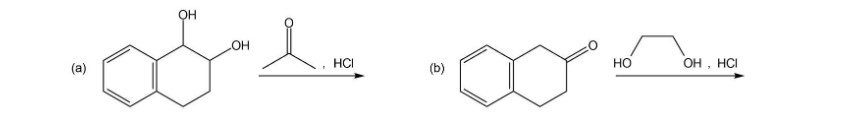
What do you think about this solution?
We value your feedback to improve our textbook solutions.