Chapter 17: Q26P (page 823)
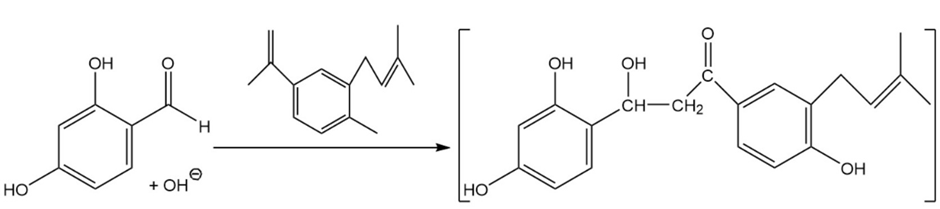
What two carbonyl compounds are required for the synthesis of morachalcone A (the aromatase inhibitor discussed in the box below), via a Claisen–Schmidt condensation?
Short Answer
The product followed as:
Learning Materials
Features
Discover
Chapter 17: Q26P (page 823)
What two carbonyl compounds are required for the synthesis of morachalcone A (the aromatase inhibitor discussed in the box below), via a Claisen–Schmidt condensation?
The product followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products of the following reactions:
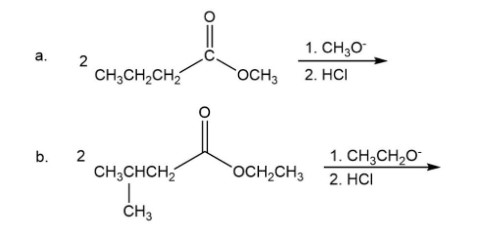
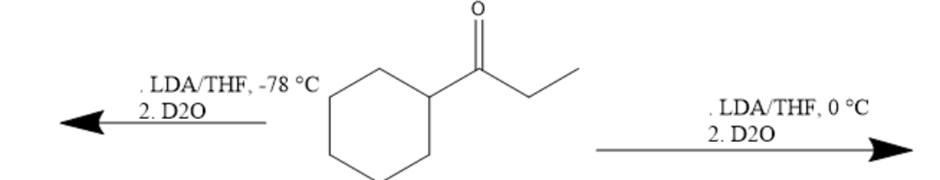
Draw the products of the following reactions:
Propose a mechanism for the formation of fructose-1, 6-bisphosphate from dihydroxyacetone phosphate and glyceraldehyde-3-phosphate, using hydroxide ion as the catalyst.
If the preference for formation of a six-membered ring were not so great, what other cyclic product would be formed from the intramolecular aldol addition of
a.2,6-heptanedione? b. 2,8-nonanedione?
a. If the biosynthesis of palmitic acid were carried out with and nondeuterated malonyl thioester, how many deuterium atoms would be incorporated into palmitic acid?
b. If the biosynthesis of palmitic acid were carried out with and nondeuterated acetyl thioester, how many deuterium atoms would be incorporated into palmitic acid?
What do you think about this solution?
We value your feedback to improve our textbook solutions.