Chapter 17: Q24P (page 820)
How could you prepare the following compound using a starting material that contains no more than three carbons?
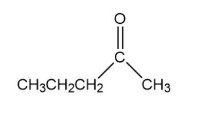
Short Answer
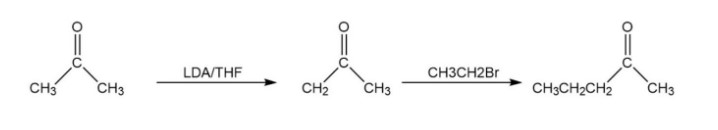
A reaction followed as:
Learning Materials
Features
Discover
Chapter 17: Q24P (page 820)
How could you prepare the following compound using a starting material that contains no more than three carbons?

A reaction followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
Using cyclopentanone as the reactant, show the product of a. acid-catalyzedketo–enolinterconversion. b. an aldol addition. c. an aldol condensation.
There are other condensation reactions similar to the aldol and claisen condensations:
a. The Perkin condensation is the condensation of an aromatic aldehyde and acetic anhydride. Draw the product obtained from the following Perkin condensation:
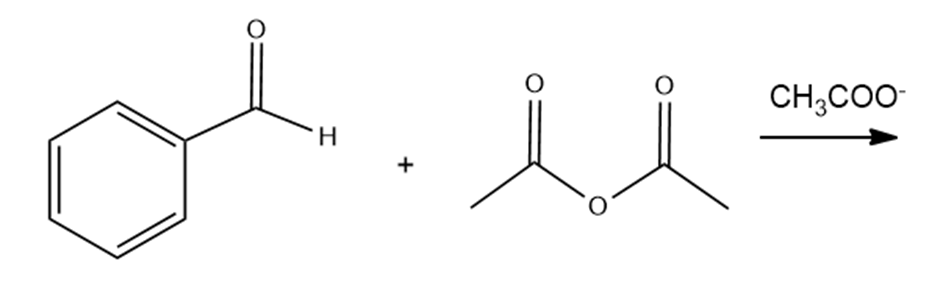
b. What compound is formed if water is added to the product of a Perkin condensation?
c. The Knoevenagel condensation is the condensation of an aldehyde or a ketone that has no alpha- hydrogens and a compound such as diethyl malonate that has an alpha-carbon flanked by two electron- withdrawing groups. Draw the product obtained from the following Knoevenagel condensation:
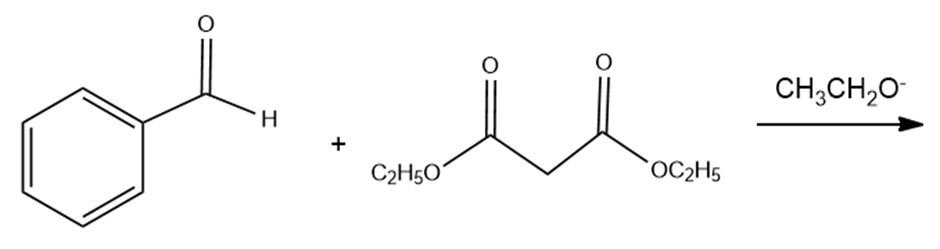
d. What product is obtained when the product of a Knoevenagel condensation is heated in an aqueous acidic solution?
Propose a mechanism for the formation of fructose-1, 6-bisphosphate from dihydroxyacetone phosphate and glyceraldehyde-3-phosphate, using hydroxide ion as the catalyst.
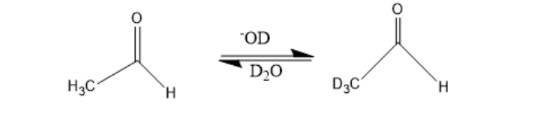
When a dilute solution of acetaldehyde in D2O containing NaOD is shaken, explain why the methyl hydrogens are exchanged with deuterium but the hydrogen attached to the carbonyl carbon is not.
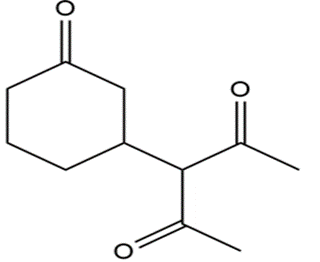
What reagents should be used to prepare the following compounds?
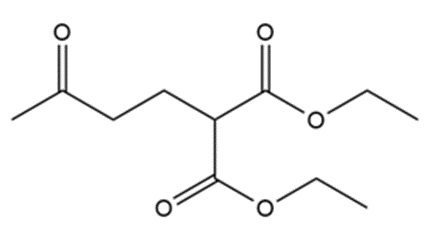
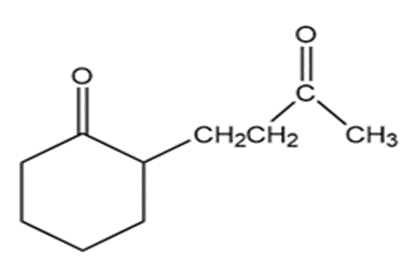
What do you think about this solution?
We value your feedback to improve our textbook solutions.