Chapter 17: Q44P (page 838)
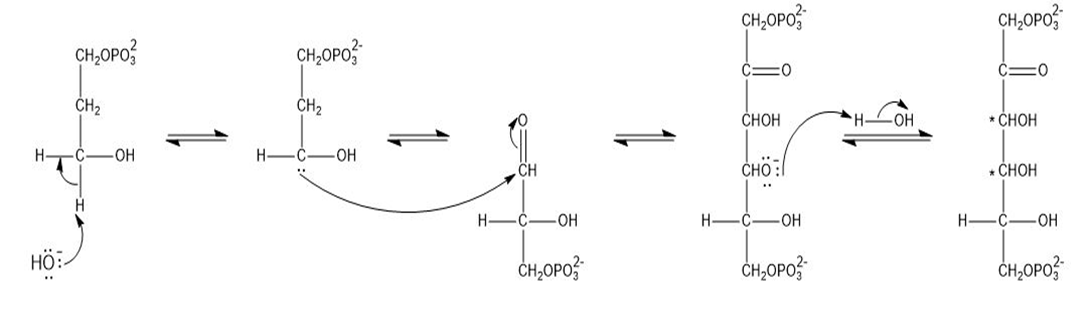
Propose a mechanism for the formation of fructose-1, 6-bisphosphate from dihydroxyacetone phosphate and glyceraldehyde-3-phosphate, using hydroxide ion as the catalyst.
Short Answer
Learning Materials
Features
Discover
Chapter 17: Q44P (page 838)
Propose a mechanism for the formation of fructose-1, 6-bisphosphate from dihydroxyacetone phosphate and glyceraldehyde-3-phosphate, using hydroxide ion as the catalyst.

All the tools & learning materials you need for study success - in one app.
Get started for free
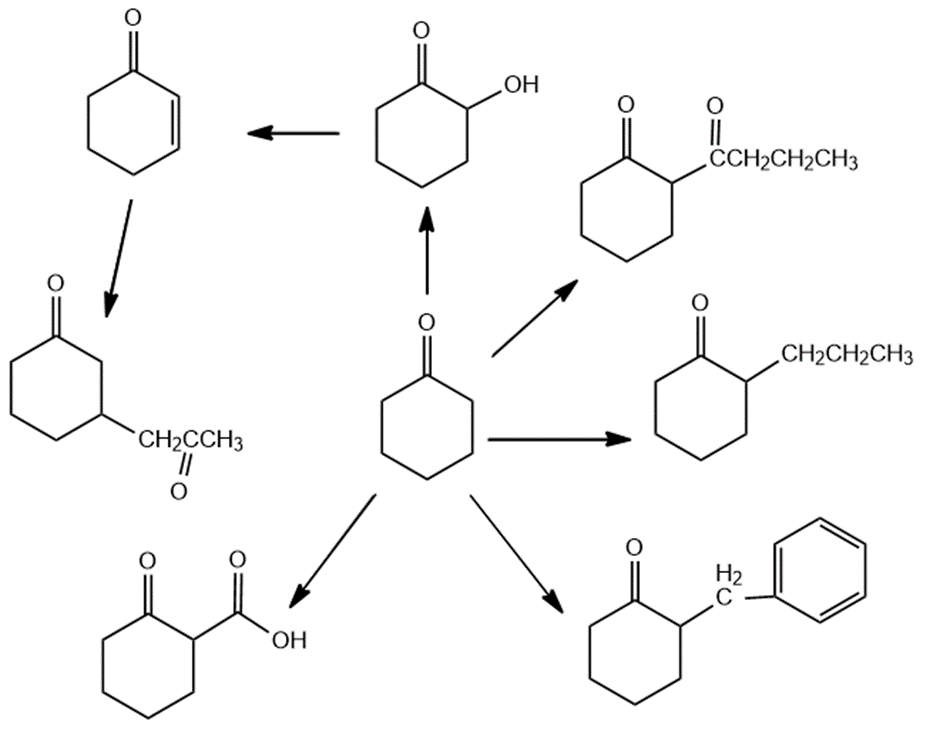
Show how the following compounds can be prepared from cyclohexanone:
Compound A with molecular formula C6H10 has two peaks in its 1H NMR spectrum, both of which are singlets (with ratio 9 : 1). Compound Areacts
with an acidic aqueous solution containing mercuric sulfate to form compound B,which gives a positive iodoform test (Problem 58) and has an 1H NMR
spectrum that shows two singlets (with ratio 3 : 1). Identify A and B.
a. If the biosynthesis of palmitic acid were carried out with and nondeuterated malonyl thioester, how many deuterium atoms would be incorporated into palmitic acid?
b. If the biosynthesis of palmitic acid were carried out with and nondeuterated acetyl thioester, how many deuterium atoms would be incorporated into palmitic acid?
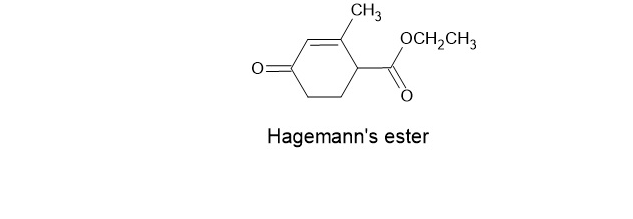
A compound known as Hagemann’s estercan be prepared by treating a mixture of formaldehyde and ethyl acetoacetate first with base and then with acid
and heat. Write the structure for the product of each of the steps.
a. The first step is an aldol-like condensation.
b. The second step is a Michael addition.
c. The third step is an intramolecular aldol addition.
d. The final transformation includes a dehydration and a hydrolysis followed by a decarboxylation.
Indicate how the following compounds can be synthesized from cyclohexanone and any other necessary reagents:
What do you think about this solution?
We value your feedback to improve our textbook solutions.