Chapter 17: Q19P (page 815)
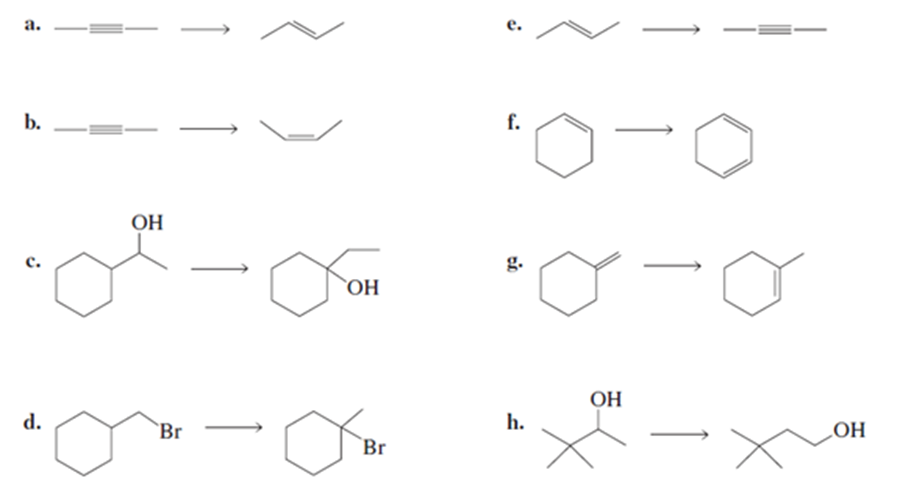
What reagents should be used to prepare the following compounds?
Learning Materials
Features
Discover
Chapter 17: Q19P (page 815)
What reagents should be used to prepare the following compounds?
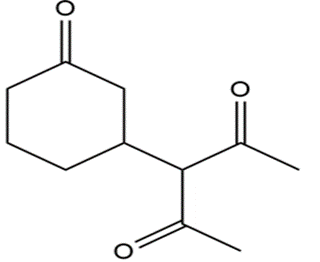
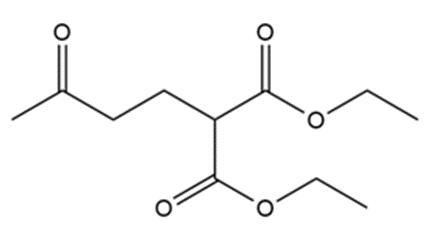
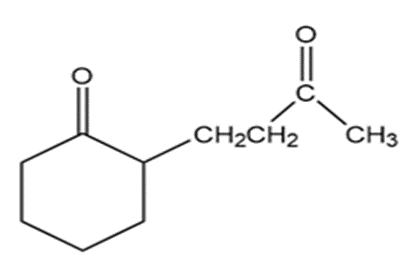
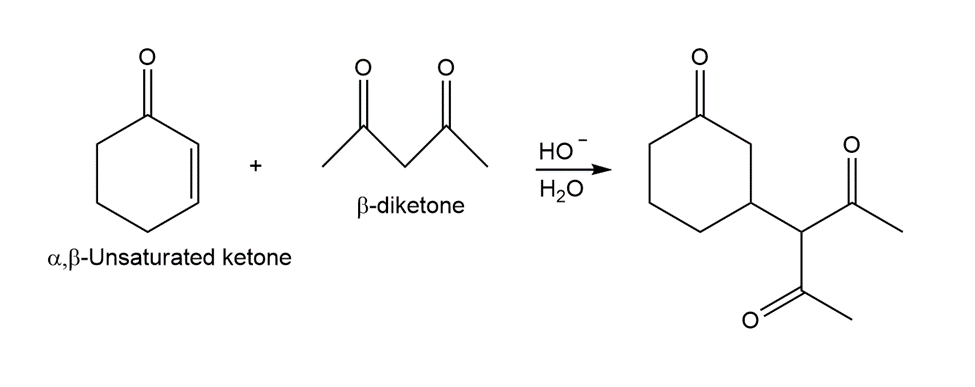
All the tools & learning materials you need for study success - in one app.
Get started for free
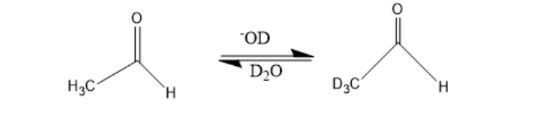
When a dilute solution of acetaldehyde in D2O containing NaOD is shaken, explain why the methyl hydrogens are exchanged with deuterium but the hydrogen attached to the carbonyl carbon is not.
Can 2,4-pentanedione undergo an intramolecular aldol addition? If so, why? If not, why not?
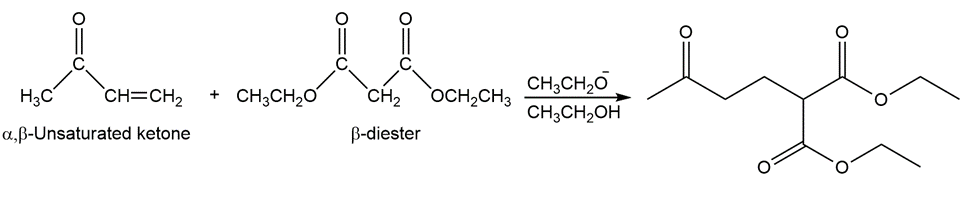
What two carbonyl compounds are required for the synthesis of morachalcone A (the aromatase inhibitor discussed in the box below), via a Claisen–Schmidt condensation?
Problem:What reagents are required to convert the reactant to the product?
Explain why alkylation of anα α -carbon works best if the alkyl halide used in the reaction is a primary alkyl halide, and why alkylation does not work at all if a tertiary alkyl halide is used.
What do you think about this solution?
We value your feedback to improve our textbook solutions.