Chapter 17: 17-55P (page 847)
Draw the products of the following reactions:
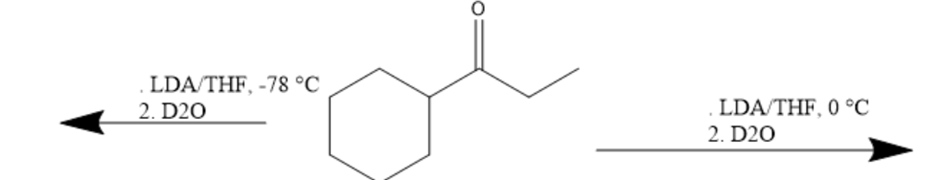
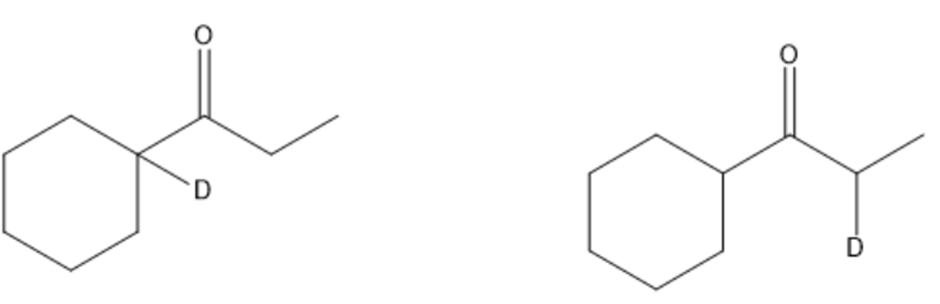
Short Answer
Learning Materials
Features
Discover
Chapter 17: 17-55P (page 847)
Draw the products of the following reactions:


All the tools & learning materials you need for study success - in one app.
Get started for free
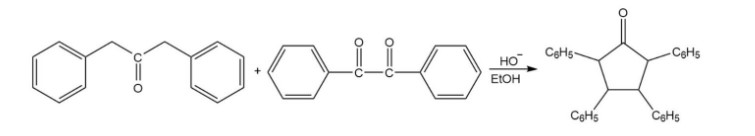
Propose a mechanism for the following reaction:
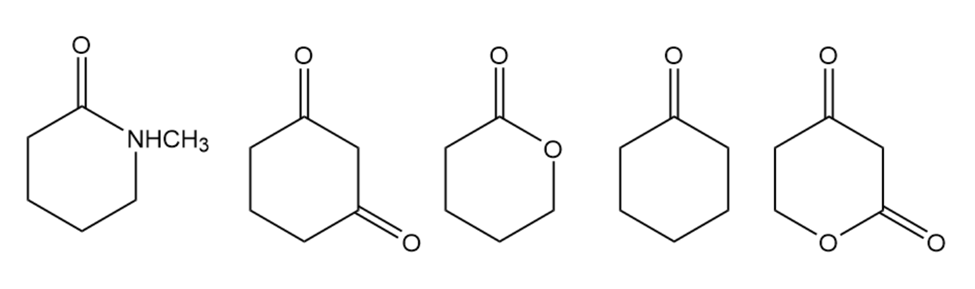
Number the following compounds in order of increasing pKa value.
Draw the products of the following reactions:
a. diethyl heptanedioate: (1) sodium ethoxide; (2) HCl
b. pentanoic acid + PBr3 + Br2, followed by hydrolysis
c. acetone + LDA/THF: (1) slow addition of ethyl acetate; (2) HCl
d. diethyl 2-ethylhexanedioate: (1) sodium ethoxide; (2) HCl
e. diethyl malonate: (1) sodium ethoxide; (2) isobutyl bromide; (3) HCl, H2O + ∆
f. acetophenone + LDA/THF: (1) slow addition of diethyl carbonate; (2) HCl
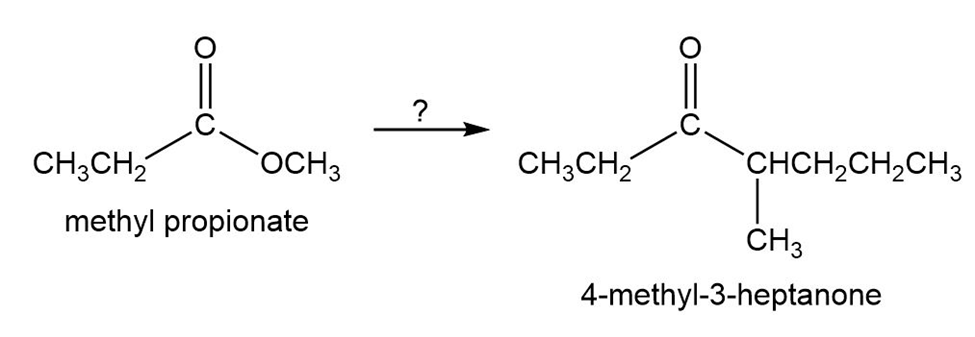
Starting with methyl propanoate, how could you prepare 4-methyl-3-heptanone?
A racemic mixture of 2-methyl-1-phenyl-1-butanone is formed when (R)-2-methyl-1-phenyl-1-butanone is dissolved in an acidic or basic aqueous solution. Give an example of another ketone that undergoes acid- or base-catalyzed racemization.
What do you think about this solution?
We value your feedback to improve our textbook solutions.