Chapter 17: Q42P (page 835)
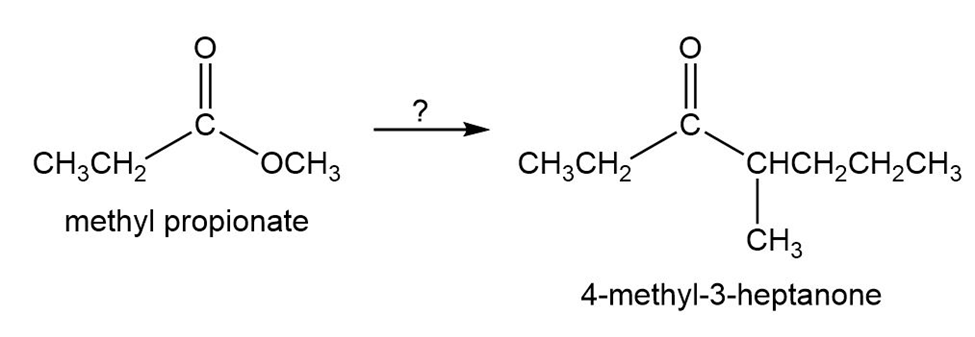
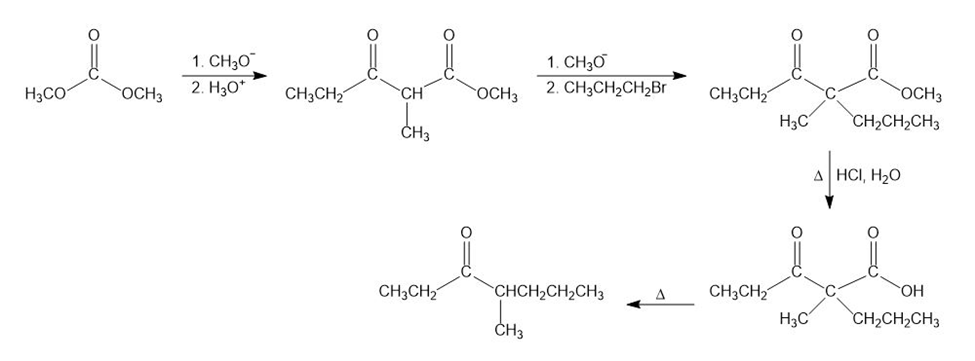
Starting with methyl propanoate, how could you prepare 4-methyl-3-heptanone?
Short Answer
Learning Materials
Features
Discover
Chapter 17: Q42P (page 835)
Starting with methyl propanoate, how could you prepare 4-methyl-3-heptanone?


All the tools & learning materials you need for study success - in one app.
Get started for free
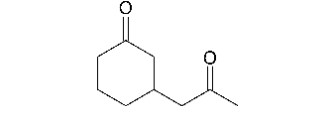
Describe how the following compound can be synthesized from compounds containing no more than six carbons.
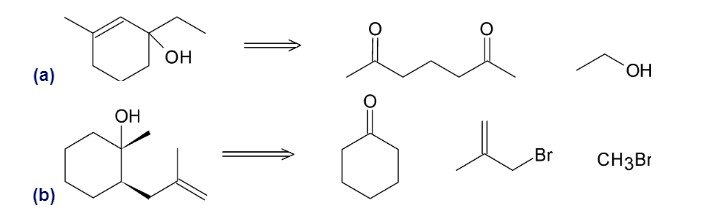
Show how the following compounds can be synthesized from the given starting materials:
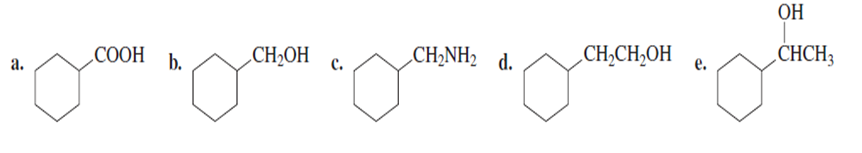
Problem: starting with bromocyclohexane, how can each of the following compounds be prepared?
Explain why alkylation of anα α -carbon works best if the alkyl halide used in the reaction is a primary alkyl halide, and why alkylation does not work at all if a tertiary alkyl halide is used.
Amobarbital is a sedative marketed under the trade name Amytal. Propose a synthesis of amobarbital, using diethyl malonate and ureaas two of the starting materials.
What do you think about this solution?
We value your feedback to improve our textbook solutions.