Chapter 17: Q41P (page 835)
What alkyl bromide should be used in the acetoacetic ester synthesis of each of the following methyl ketones?
a. 2-pentanone b. 2-octanone c. 4-phenyl-2-butanone
Short Answer
- Ethyl bromide
- Pentyl bromide
- Benzyl bromide
Learning Materials
Features
Discover
Chapter 17: Q41P (page 835)
What alkyl bromide should be used in the acetoacetic ester synthesis of each of the following methyl ketones?
a. 2-pentanone b. 2-octanone c. 4-phenyl-2-butanone
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a structure for each of the following:
a. ethyl acetoacetate
b. -methylmalonic acid
c. a -keto ester
d. the enol tautomer of cyclopentanone
e. the carboxylic acid obtained from the malonic ester synthesis when the alkyl halide is propyl bromide
Draw the products of the following reactions:
a. diethyl heptanedioate: (1) sodium ethoxide; (2) HCl
b. pentanoic acid + PBr3 + Br2, followed by hydrolysis
c. acetone + LDA/THF: (1) slow addition of ethyl acetate; (2) HCl
d. diethyl 2-ethylhexanedioate: (1) sodium ethoxide; (2) HCl
e. diethyl malonate: (1) sodium ethoxide; (2) isobutyl bromide; (3) HCl, H2O + ∆
f. acetophenone + LDA/THF: (1) slow addition of diethyl carbonate; (2) HCl
a. If the biosynthesis of palmitic acid were carried out with and nondeuterated malonyl thioester, how many deuterium atoms would be incorporated into palmitic acid?
b. If the biosynthesis of palmitic acid were carried out with and nondeuterated acetyl thioester, how many deuterium atoms would be incorporated into palmitic acid?
A racemic mixture of 2-methyl-1-phenyl-1-butanone is formed when (R)-2-methyl-1-phenyl-1-butanone is dissolved in an acidic or basic aqueous solution. Give an example of another ketone that undergoes acid- or base-catalyzed racemization.
Problem:What reagents are required to convert the reactant to the product?
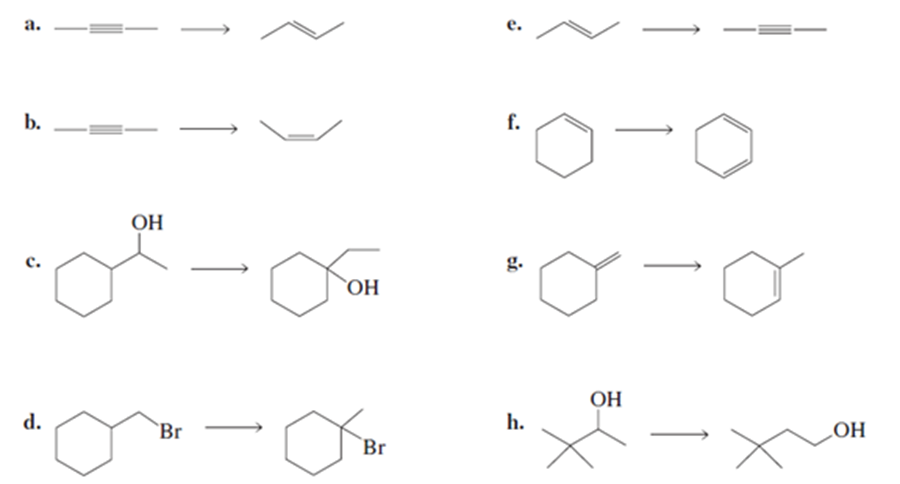
What do you think about this solution?
We value your feedback to improve our textbook solutions.