Chapter 14: Q25P (page 642)
How can theirNMR spectra distinguish the following compounds?
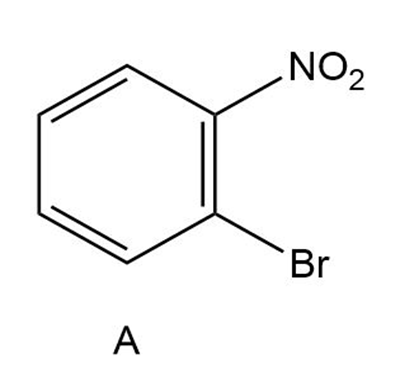
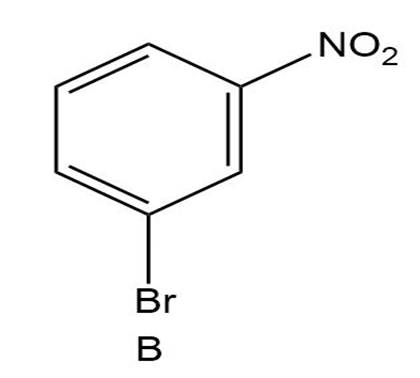
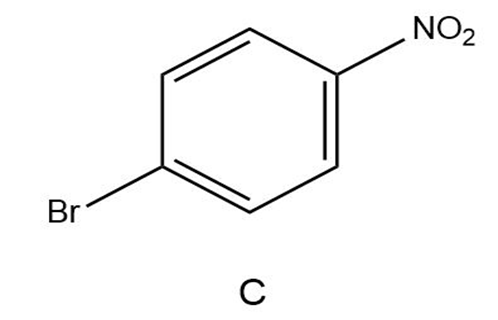
Short Answer
A)
B)
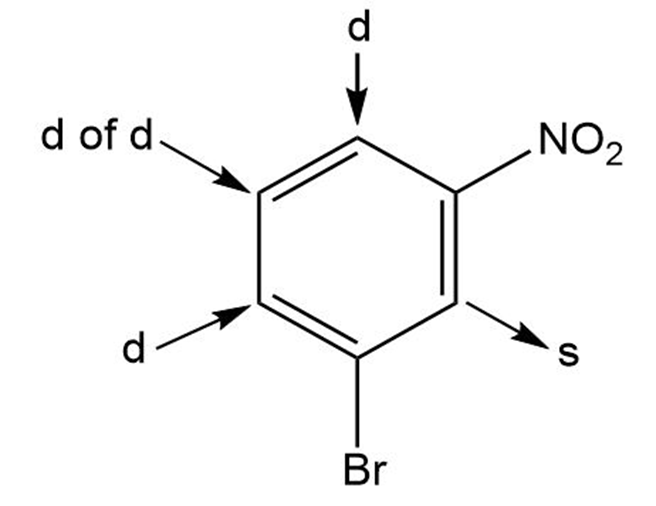
C)
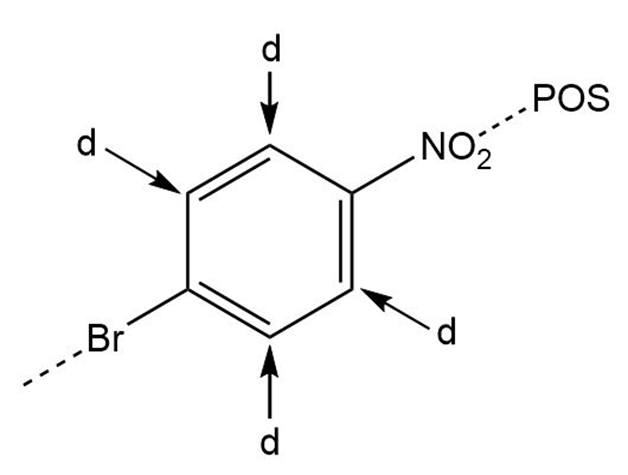
Learning Materials
Features
Discover
Chapter 14: Q25P (page 642)
How can theirNMR spectra distinguish the following compounds?



A)

B)

C)

All the tools & learning materials you need for study success - in one app.
Get started for free
Determine the ratios of the chemically non-equivalent protons in a compound if the steps of the integration curves measure and , from left to right across the spectrum. Draw the structure of a compound whose NMR spectrum would show these integrals in the observed order.
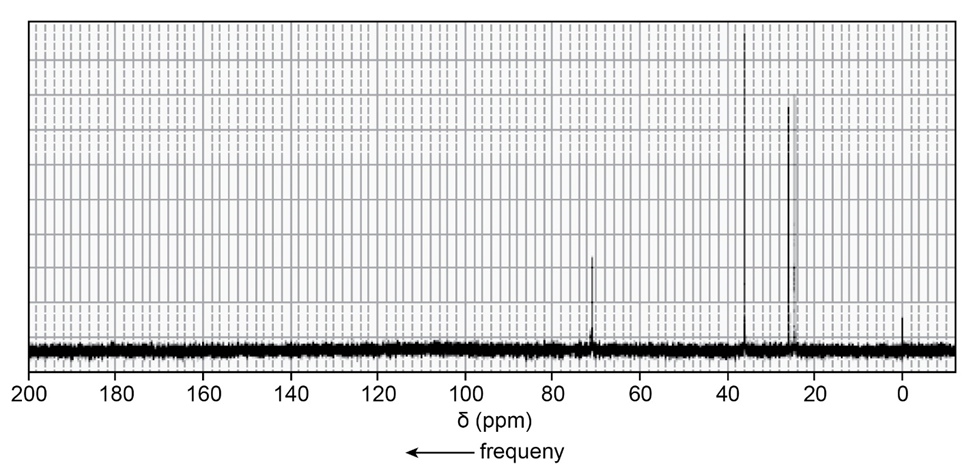
Identify each of the following compounds from its molecular formula and its 13C NMR spectrum:
a.C4H10O
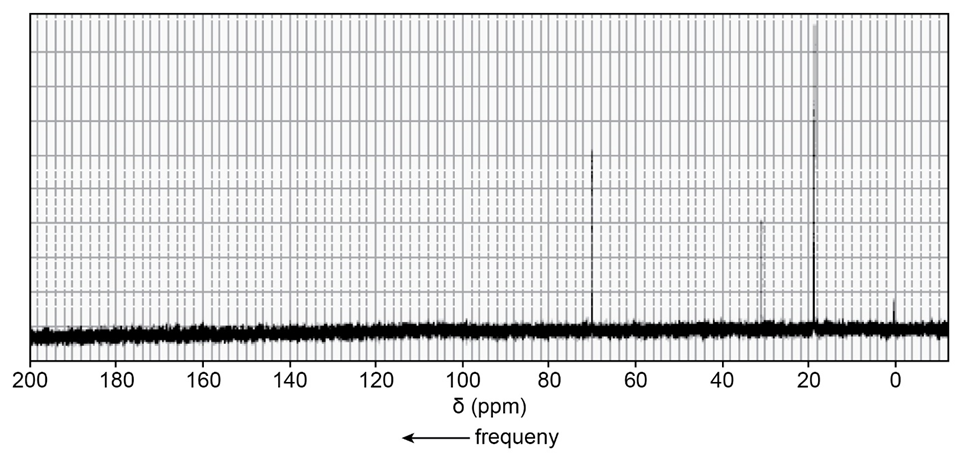
b.C6H12O
Why is there no coupling between the a and c protons or between the b and c protons in the cis and trans alkenes shown in Figure 14.20?
Draw a splitting diagram for the proton and give its multiplicity if
a. b.
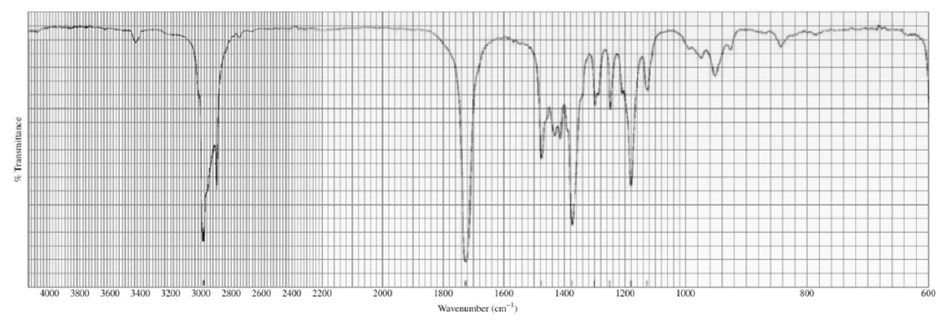
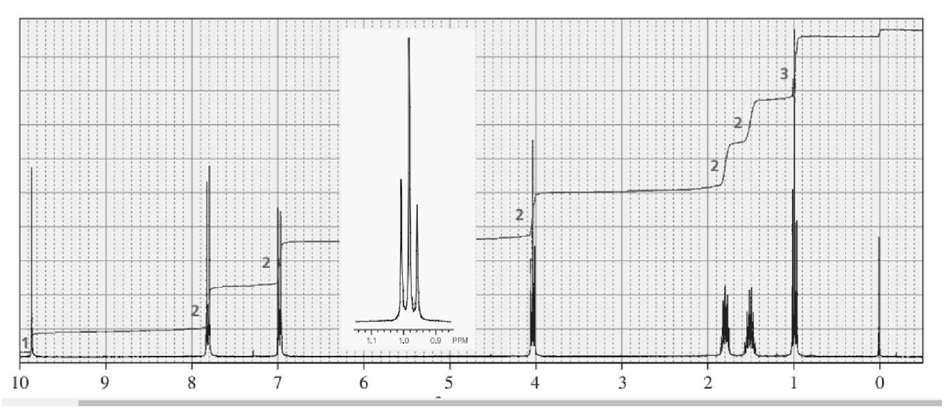
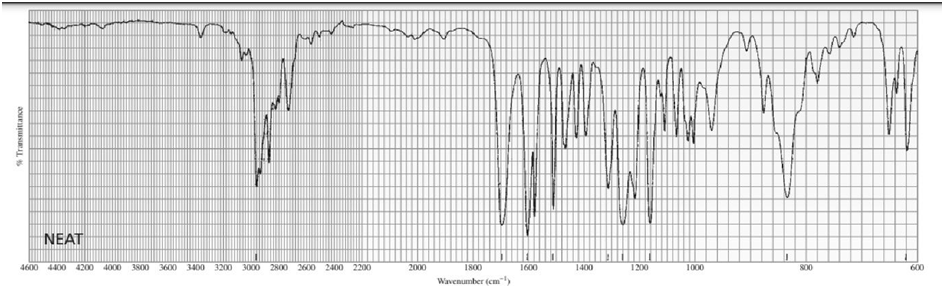
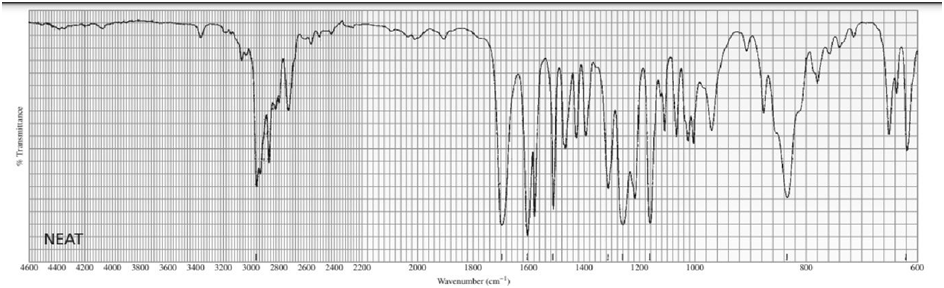
Identify each of the following compounds from its molecular formula and its IR and 1H NMR spectra:
a. C6H120
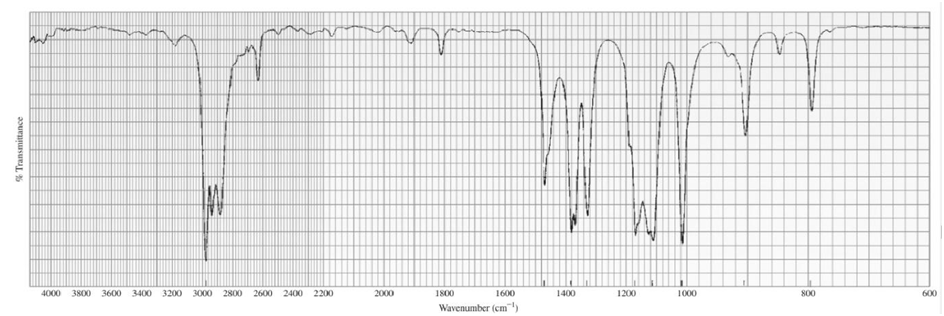
b.C6H14O
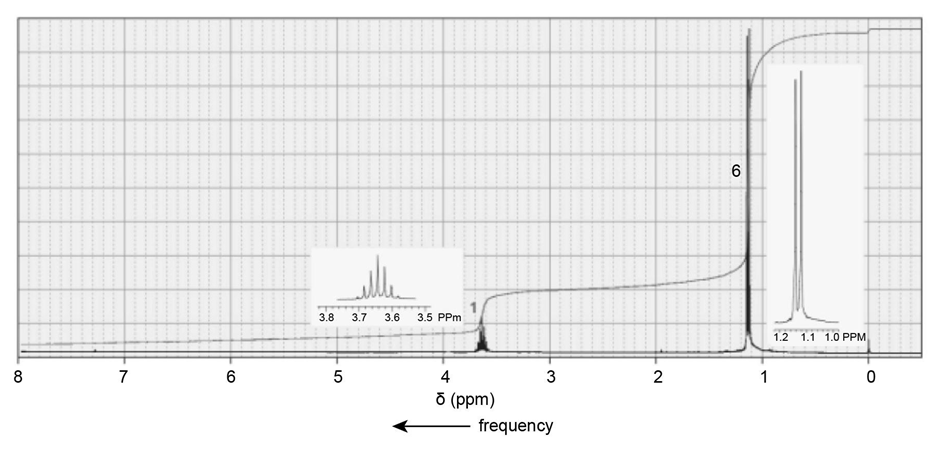
c. C10H13NO3
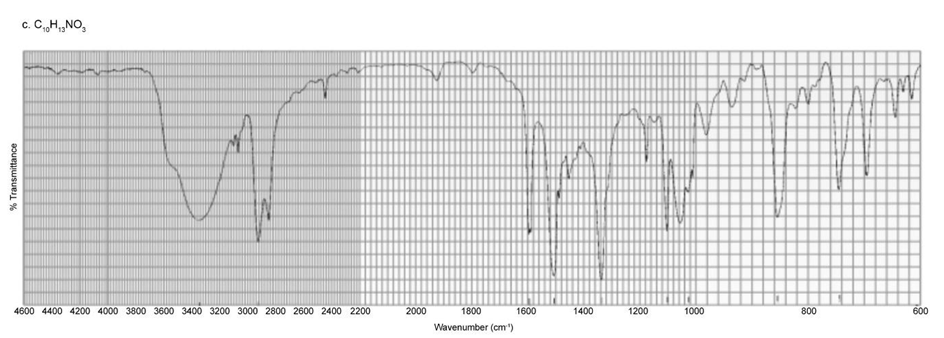
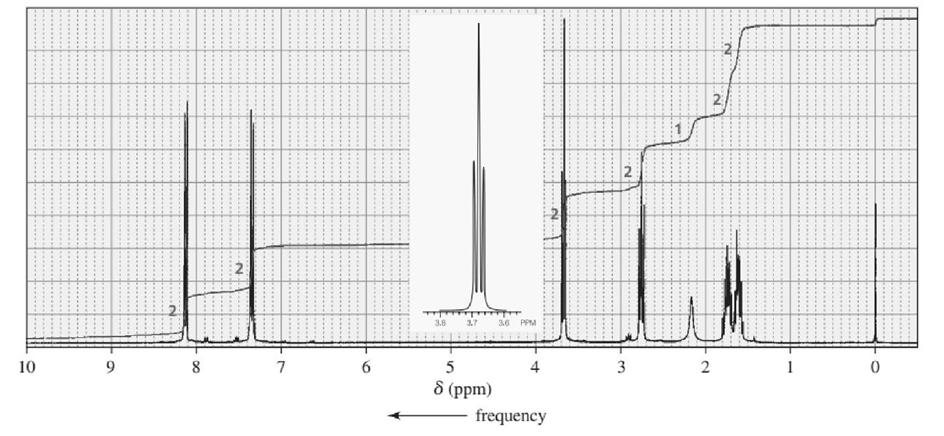
d. C11H14O2
What do you think about this solution?
We value your feedback to improve our textbook solutions.