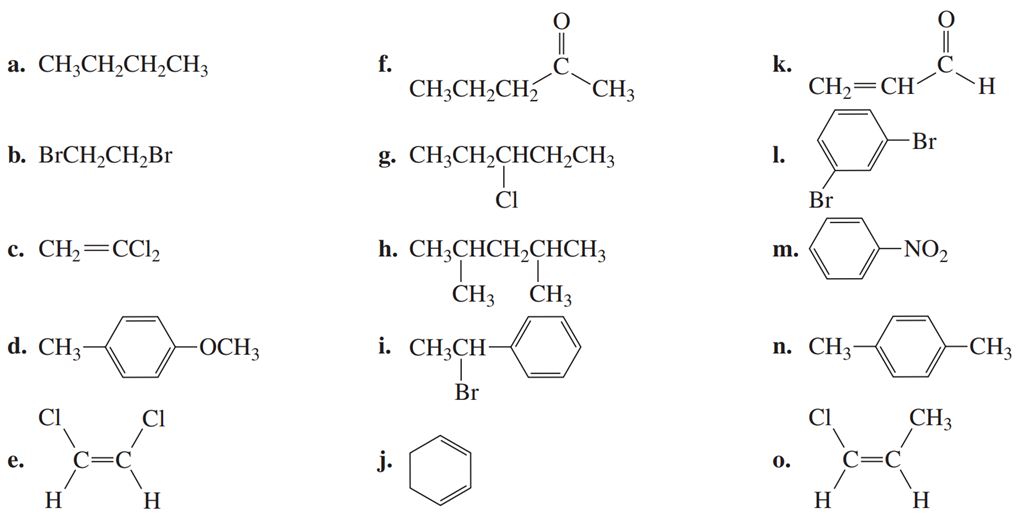
Chapter 14: Q50P (page 669)
Determine the ratios of the chemically non-equivalent protons in a compound if the steps of the integration curves measure and , from left to right across the spectrum. Draw the structure of a compound whose NMR spectrum would show these integrals in the observed order.
Short Answer
Ratio of hydrogen atoms is and the structure of the compound is given below.
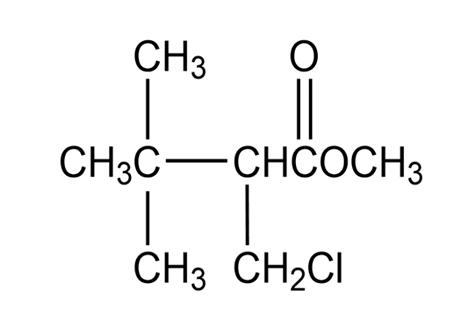
 Structure of desired compound
Structure of desired compound