Chapter 14: Q48P (page 669)
Draw a splitting diagram for the proton and give its multiplicity if
a. b.
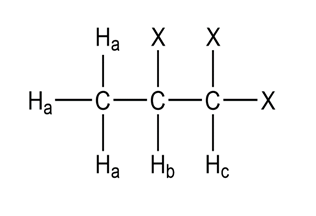
Short Answer
a.
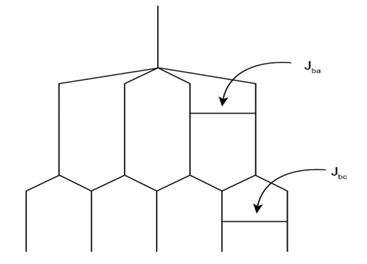
This has five peaks, called quintet.
b.
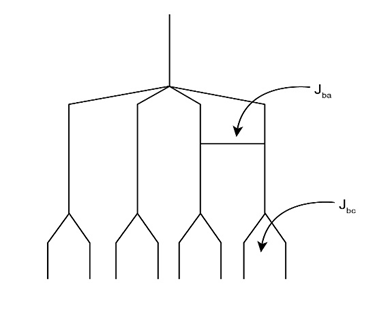
This has eight peaks i.e., double of quartet.
Learning Materials
Features
Discover
Chapter 14: Q48P (page 669)
Draw a splitting diagram for the proton and give its multiplicity if
a. b.

a.

This has five peaks, called quintet.
b.

This has eight peaks i.e., double of quartet.
All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the splitting patterns for the signals given by compounds in Problem 4.
a. If two signals differ by 1.5 ppm in a 300 MHz spectrometer, by how much do they differ in a 500 MHz spectrometer?
b. If two signals differ by 90 Hz in a 300 MHz spectrometer, by how much do they differ in a 500 MHz spectrometer?
Dr. N. M. Arr was called in to help analyze the 1 H NMR spectrum of a mixture of compounds known to contain only C, H, and Br. The mixture showed two singlets-one at 1.8 ppm and the other at 2.7 ppm-with relative integrals of 1: 6, respectively. Dr. Arr determined that the spectrum was that of a mixture of bromomethane and 2-Bromo-2-methylpropane. What was the ratio of bromomethane to 2-Bromo-2-methylpropane in the mixture?
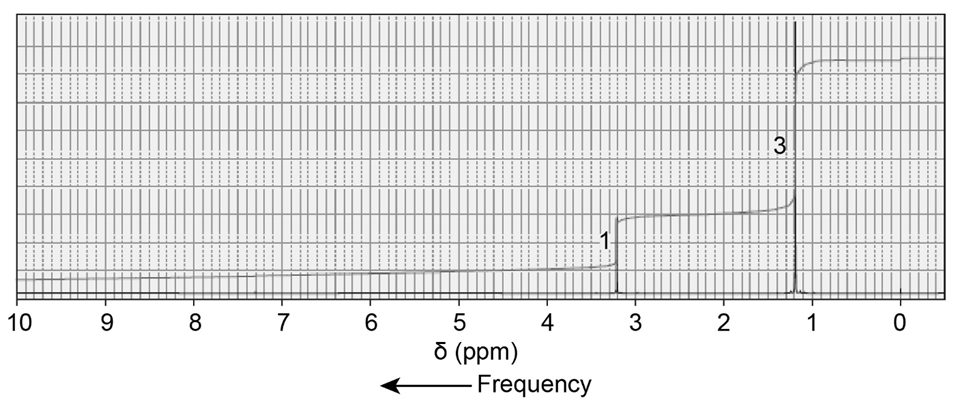
An alkyl halide reacts with an alkoxide ion to form a compound whose 1H NMR spectrum is shown here. Identify the alkyl halide and the alkoxide ion.
Draw a diagram like the one shown in Figure 14.12 to predict;
a. the relative intensities of the peaks in a triplet.
b. the relative intensities of the peaks in a quintet.
What do you think about this solution?
We value your feedback to improve our textbook solutions.