Chapter 14: Q27P (page 643)
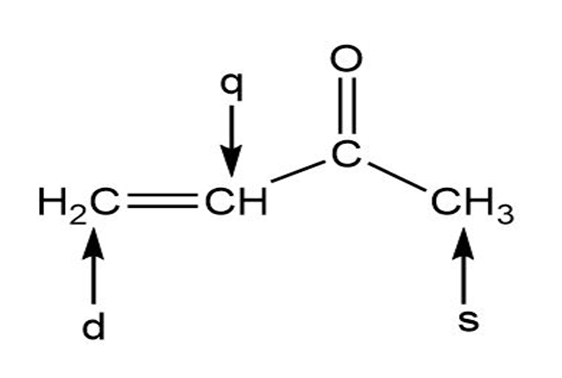
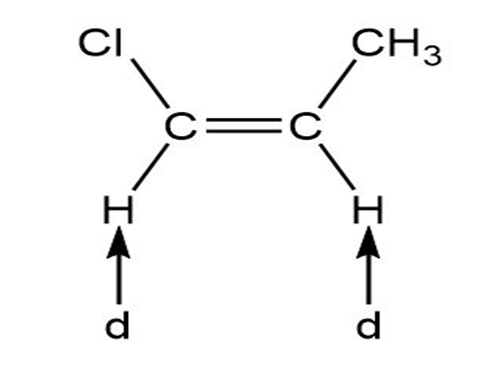
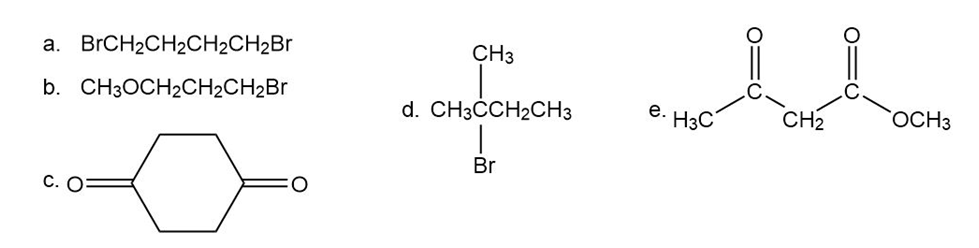
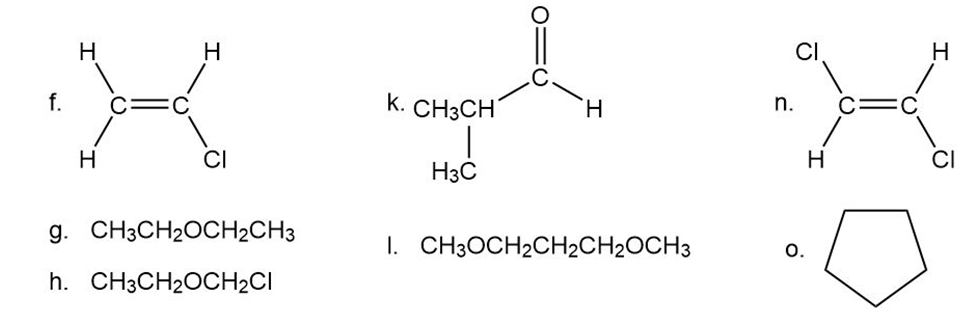
Predict the splitting patterns for the signals given by compounds in Problem 4.
Short Answer
a.
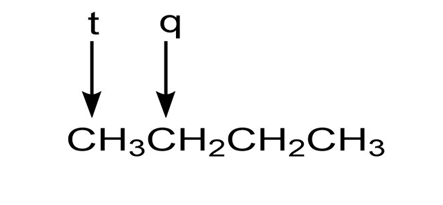
b.
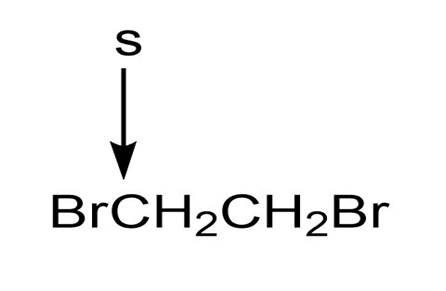
c.
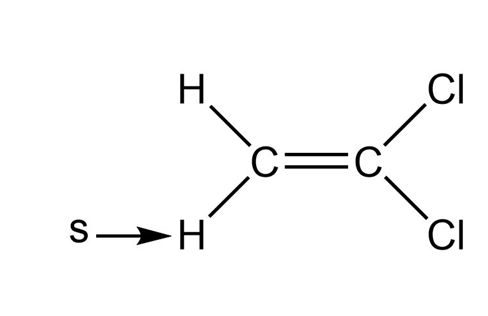
d.
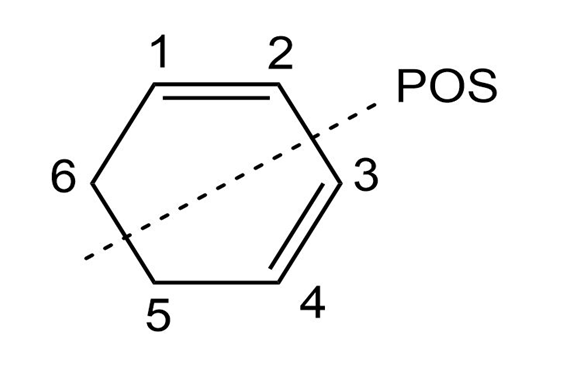
C-2,C-3,C-5,C-6 Shows doublet, while C-1, C-4 shows multiplet
e.
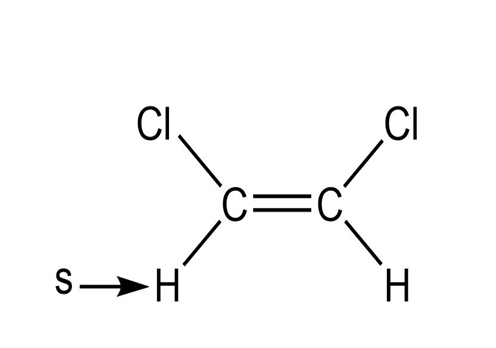
f.
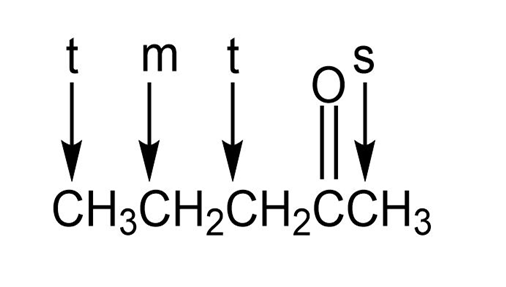
g.
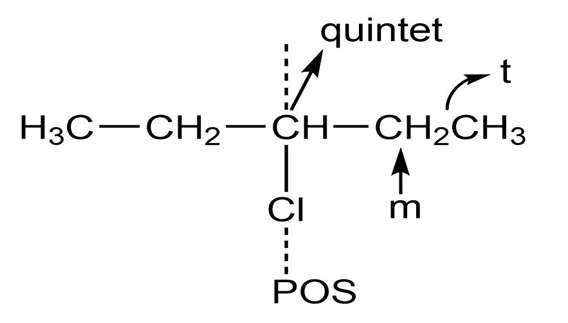
h.
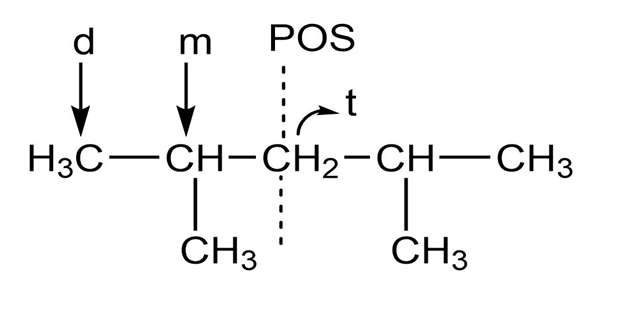
i.
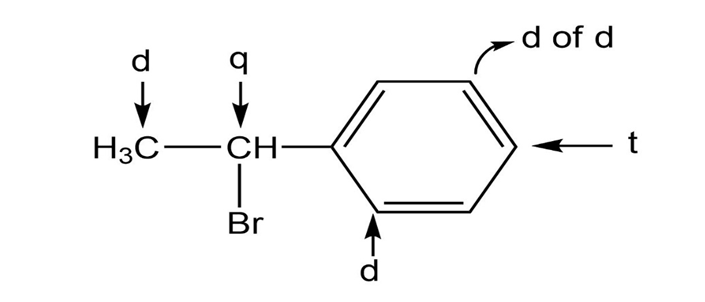
j.
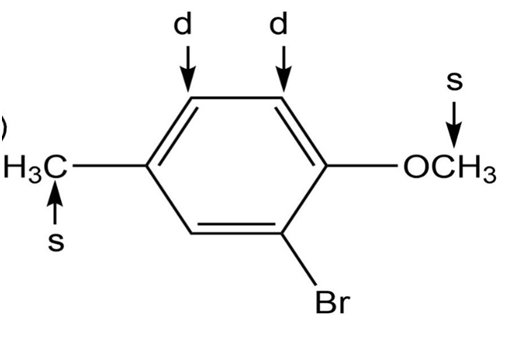
k.
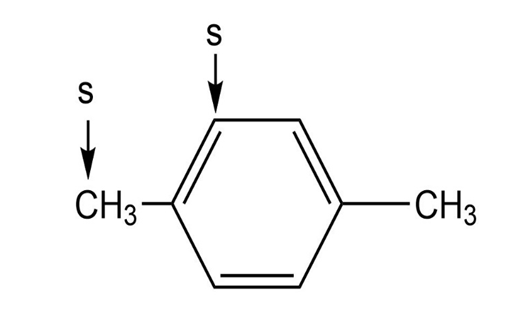
l.

m.
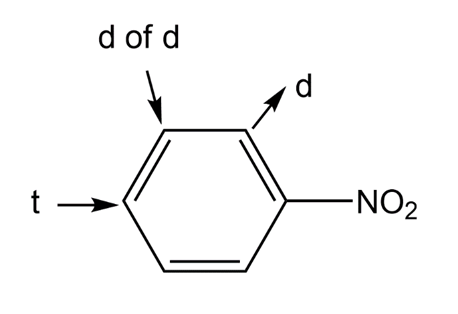
n.
o.
Learning Materials
Features
Discover
Chapter 14: Q27P (page 643)
Predict the splitting patterns for the signals given by compounds in Problem 4.
a.

b.

c.

d.

C-2,C-3,C-5,C-6 Shows doublet, while C-1, C-4 shows multiplet
e.

f.

g.

h.

i.

j.

k.

l.

m.

n.

o.

All the tools & learning materials you need for study success - in one app.
Get started for free
Describe the 1H NMR spectrum you would expect for each of the following compounds, indicating the relative positions of the signals:
Compound A, with molecular formula C4H9Cl, shows two signals in its 13C NMR spectrum. Compound B, an isomer of compound A, shows foursignals, and in the proton-coupled mode, the signal farthest downfield is a doublet. Identify compounds A and B.
When compound A ( ) is treated with HBr, it forms compound B ( ). The 1 H NMR spectrum of compound A has a 1H singlet, a 3H doublet, a 6H doublet, and two 1H multiplets. The 1 H NMR spectrum of compound B has a 6H singlet, a 3H triplet, and a 2H quartet. Identify compounds A and B.
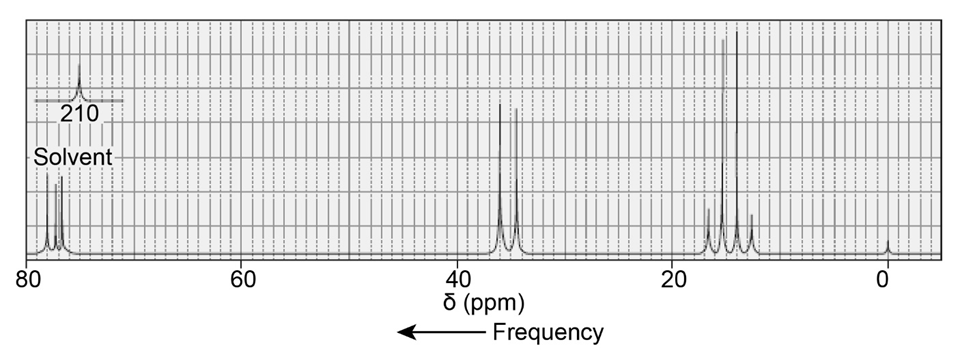
Identify the compound with molecular formula C7H14O that gives the following proton-coupled 13C NMR spectrum:
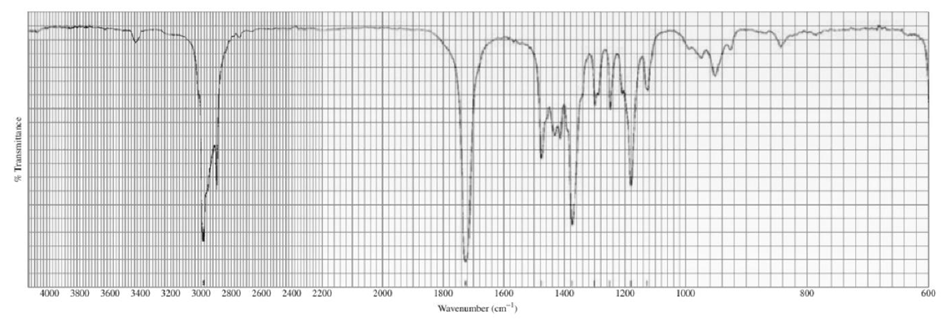
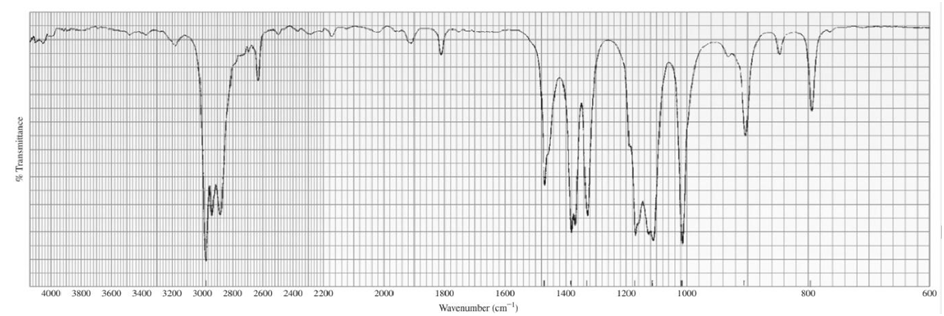
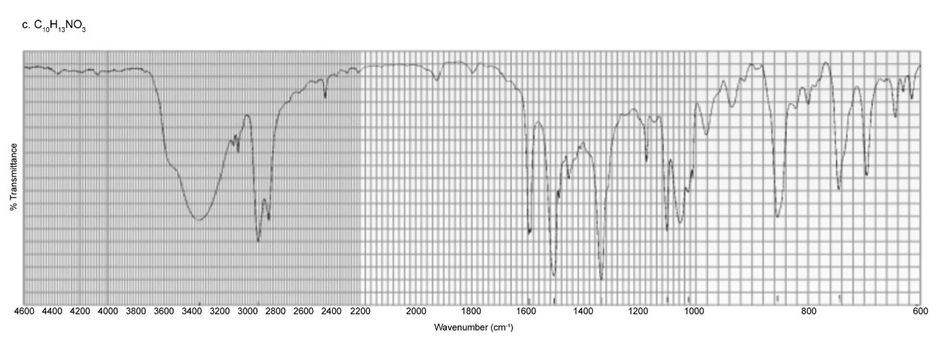
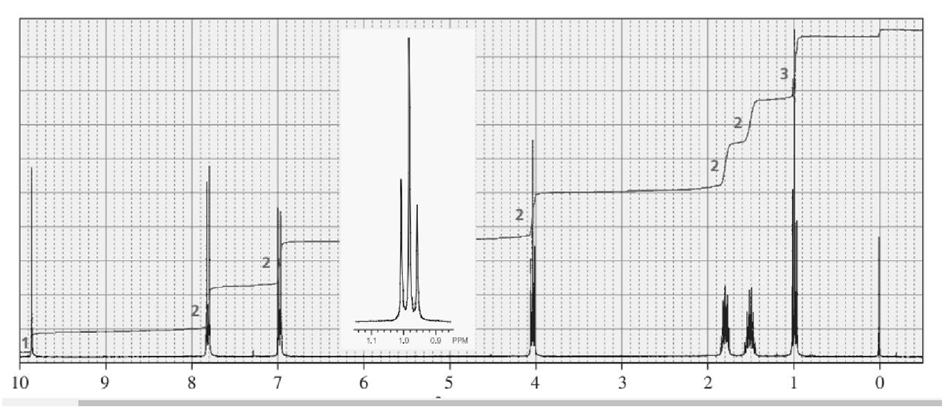
Identify each of the following compounds from its molecular formula and its IR and 1H NMR spectra:
a. C6H120
b.C6H14O
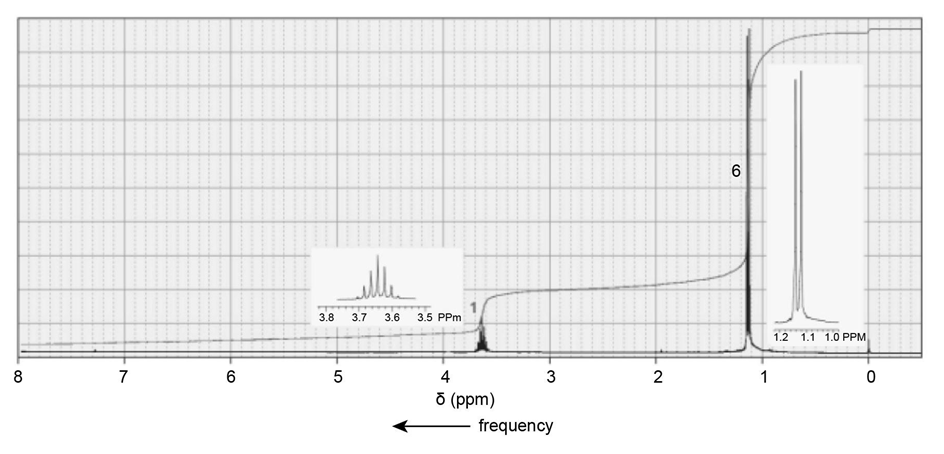
c. C10H13NO3
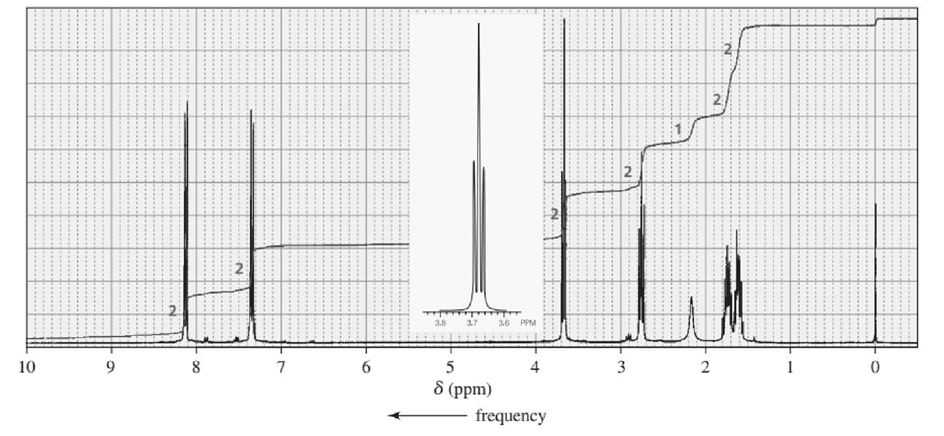
d. C11H14O2
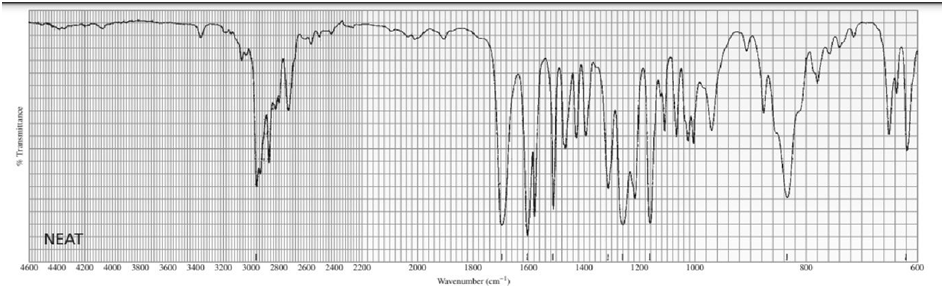
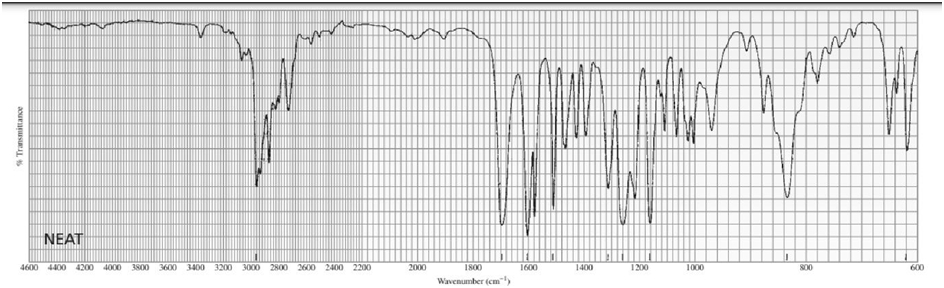
What do you think about this solution?
We value your feedback to improve our textbook solutions.