Chapter 14: Q24P (page 640)
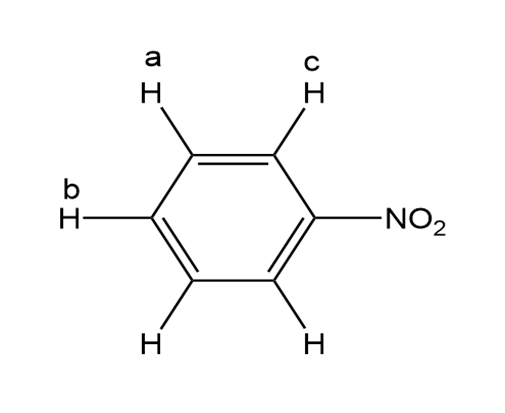
Explain the relative chemical shifts of the benzene ring protons in Figure 14.18.
Short Answer
For proton a it is 7.52 ppm.
For proton b it is 7.64 ppm.
For proton c it is 8.21 ppm.
Learning Materials
Features
Discover
Chapter 14: Q24P (page 640)
Explain the relative chemical shifts of the benzene ring protons in Figure 14.18.

For proton a it is 7.52 ppm.
For proton b it is 7.64 ppm.
For proton c it is 8.21 ppm.
All the tools & learning materials you need for study success - in one app.
Get started for free
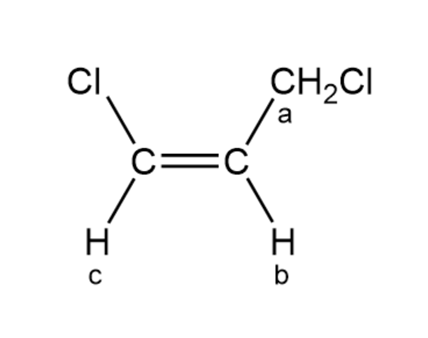
Draw a splitting diagram for the Hb proton if Jbc=10 and Jba=5.
Answer the following questions:
a. What is the relationship between chemical shift in ppm and operating frequency?
b. What is the relationship between chemical shift in hertz and operating frequency?
c. What is the relationship between coupling constant in hertz and operating frequency?
d. How does the operating frequency in NMR spectroscopy compare with the operating frequency in IR and UV/Vis spectroscopy?
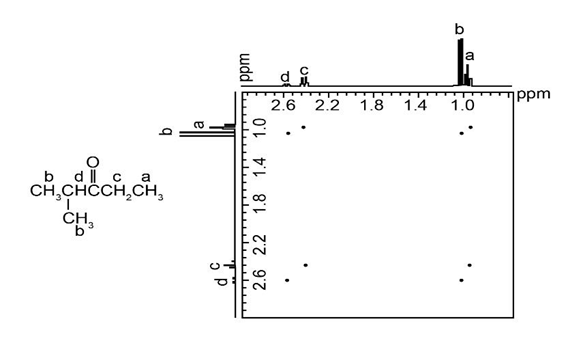
Identify pairs of coupled protons in the compound whose COSY spectrum is shown below:
Draw a diagram like the one shown in Figure 14.12 to predict;
a. the relative intensities of the peaks in a triplet.
b. the relative intensities of the peaks in a quintet.
Match each of the 1HNMR spectra with one of the following compounds:
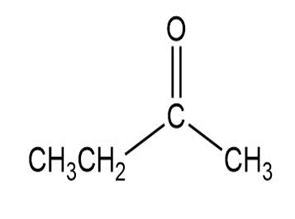
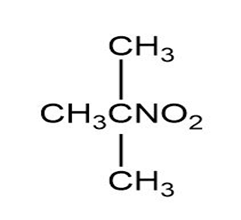
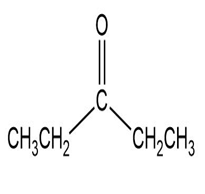
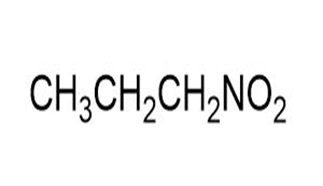
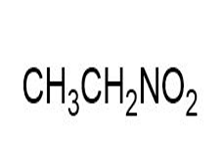
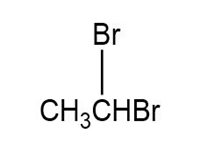
a.
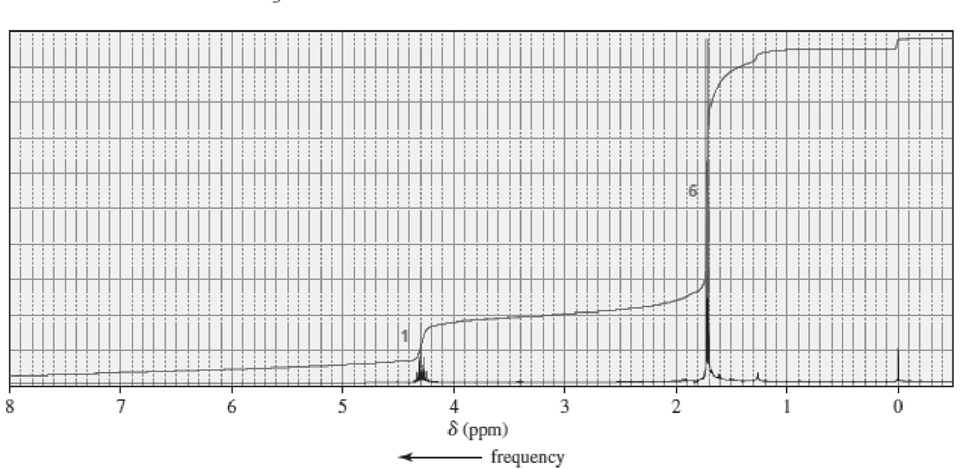
b.
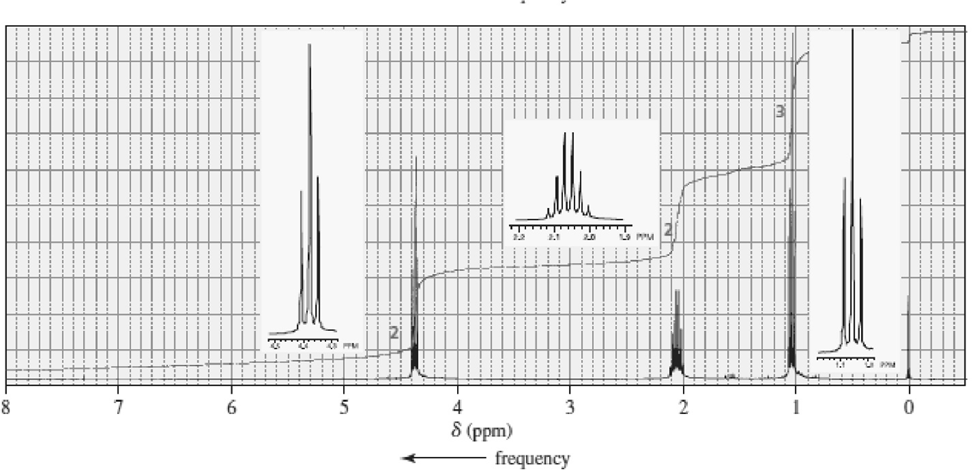
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.