Chapter 14: Q14-68P (page 677)
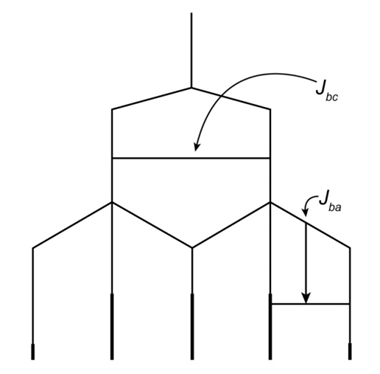
Draw a splitting diagram for the Hb proton if Jbc=10 and Jba=5.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q14-68P (page 677)
Draw a splitting diagram for the Hb proton if Jbc=10 and Jba=5.


All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for proton exchange of an alcohol in aqueous base.
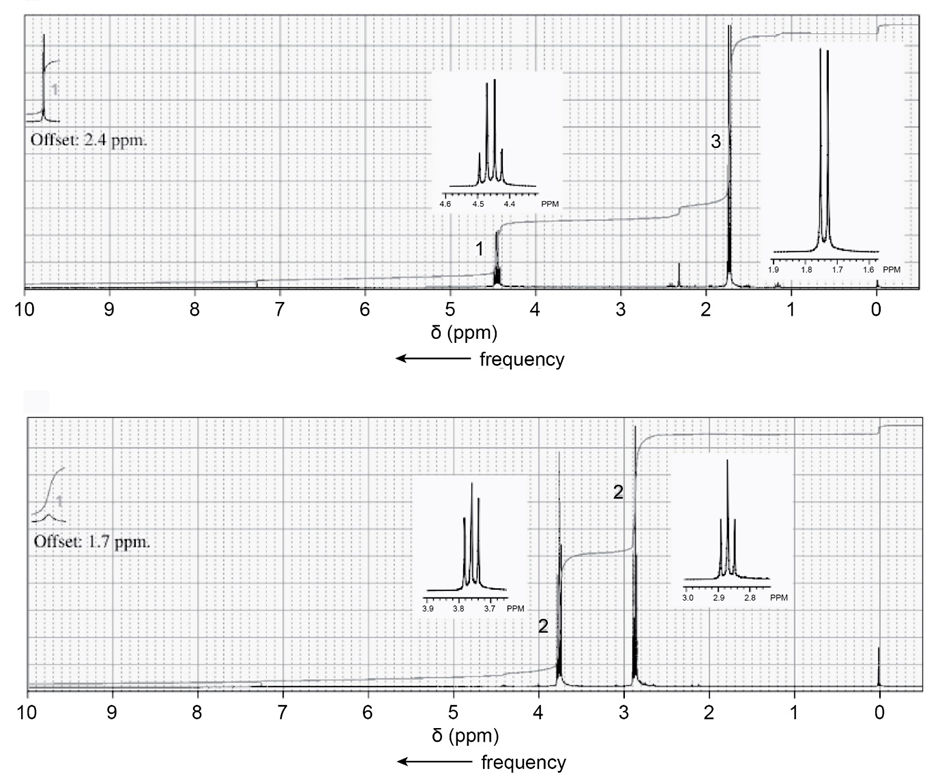
The 1 H NMR spectra of two carboxylic acids with molecular formula C3H5O2Cl are shown below. Identify the carboxylic acids. (The “offset” notation means that the farthest-left signal has been moved to the right by the indicated amount to fit on the spectrum; thus, the signal at 9.8 ppm offset by 2.4 ppm has an actual chemical shift of 9.8 + 2.4 = 12.2 ppm.)
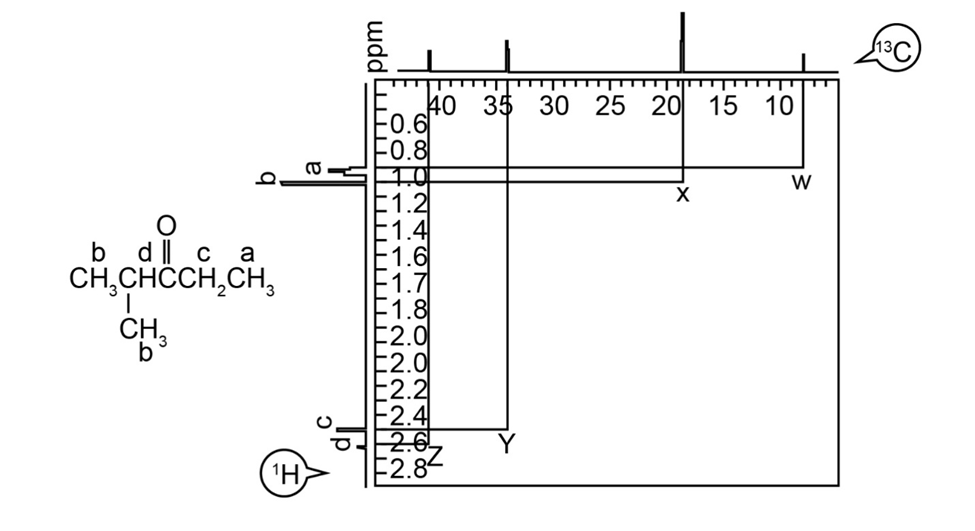
What does cross peak X in figure tell you?
A signal is seen at 600 Hz from the TMS signal in an NMR spectrometer with a 300-MHz operating frequency.
a. What is the chemical shift of the signal?
b. What is its chemical shift in an instrument operating at 500 MHz?
c. How many hertz from the TMS signal is the signal in a 500-MHz spectrometer?
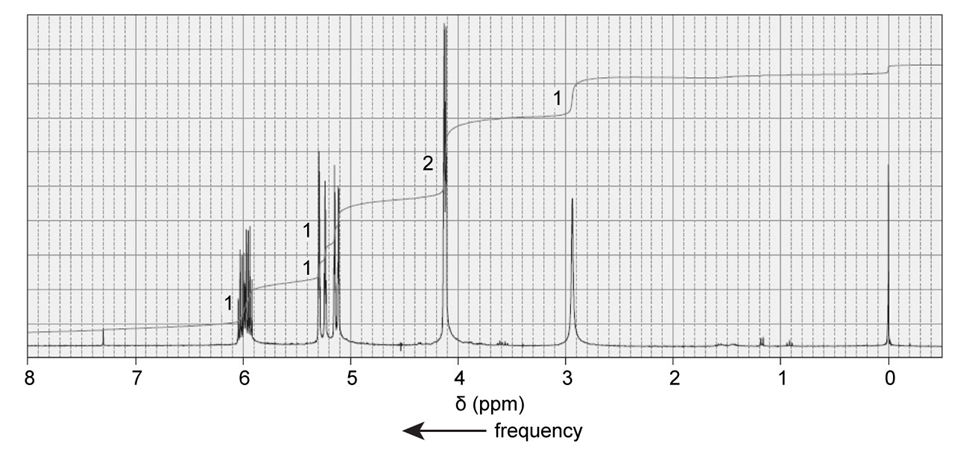
The 1H NMR spectrum of 2-propane-1-ol is shown here. Indicate the protons in the molecule that are responsible for each of the signals in the spectrum.
What do you think about this solution?
We value your feedback to improve our textbook solutions.