Chapter 14: Q14-65P (page 676)
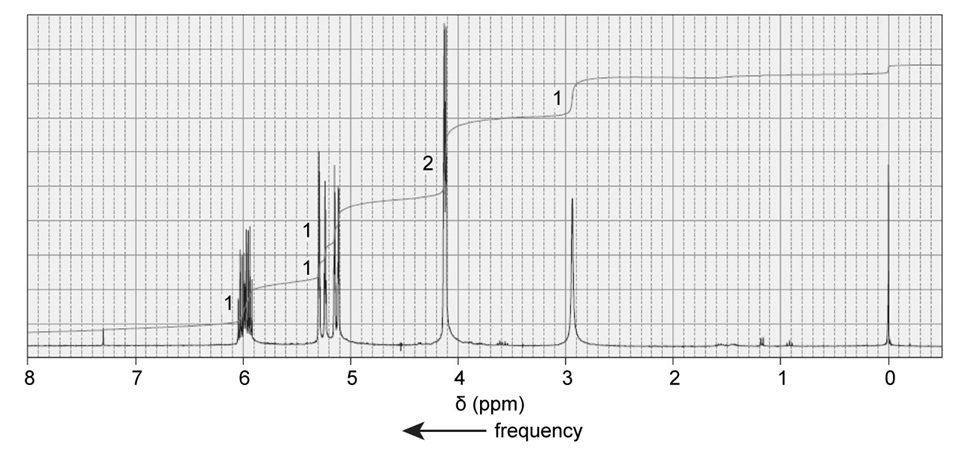
The 1H NMR spectrum of 2-propane-1-ol is shown here. Indicate the protons in the molecule that are responsible for each of the signals in the spectrum.
Short Answer
.
Learning Materials
Features
Discover
Chapter 14: Q14-65P (page 676)
The 1H NMR spectrum of 2-propane-1-ol is shown here. Indicate the protons in the molecule that are responsible for each of the signals in the spectrum.

.
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw an isomer of dichlorocyclopropane that gives anNMR spectrum
a. with one signal
b. with two signals.
c. with three signals
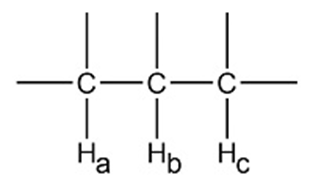
Draw a splitting diagram for Hbwere
a.Jba= 12 Hz andJbc= 6Hz b.Jba= 12 Hz andJbc= 12Hz
Draw a diagram like the one shown in Figure 14.12 to predict;
a. the relative intensities of the peaks in a triplet.
b. the relative intensities of the peaks in a quintet.
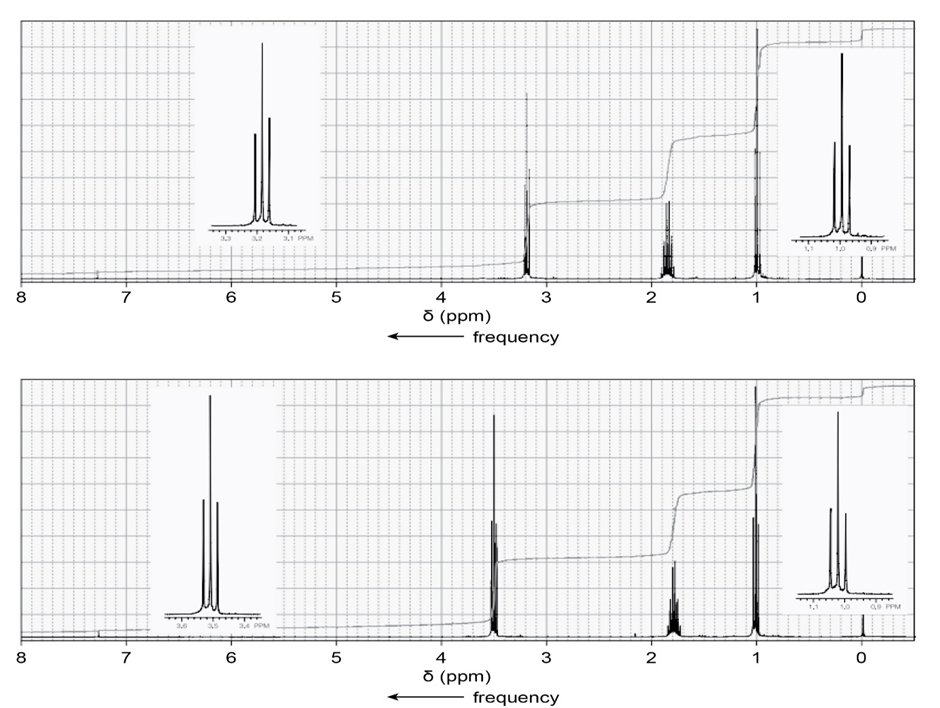
One of the spectra below is produced by 1-chloropropane and the other by 1-iodopropane. Which is which?
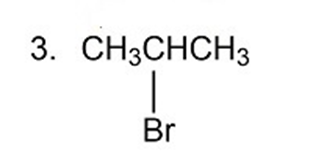
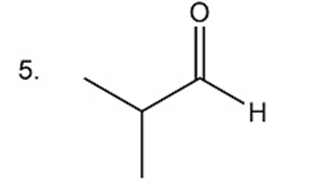
Describe the proton-coupled 13C NMR spectra for compounds 1, 3, and 5 indicating the relative positions of the signals.
What do you think about this solution?
We value your feedback to improve our textbook solutions.