Chapter 14: Q19P (page 636)
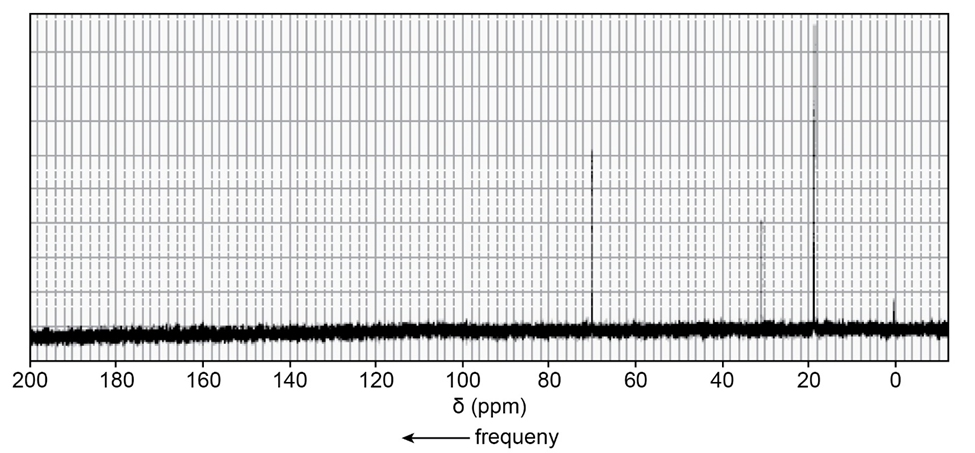
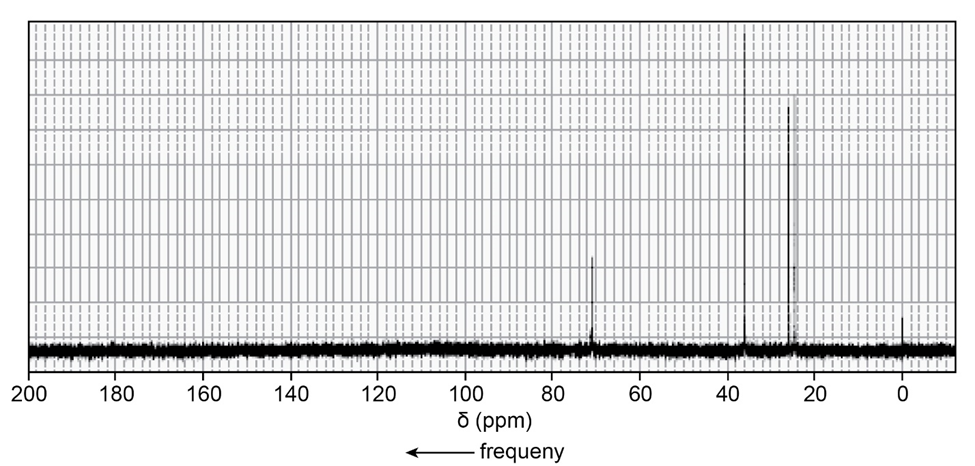
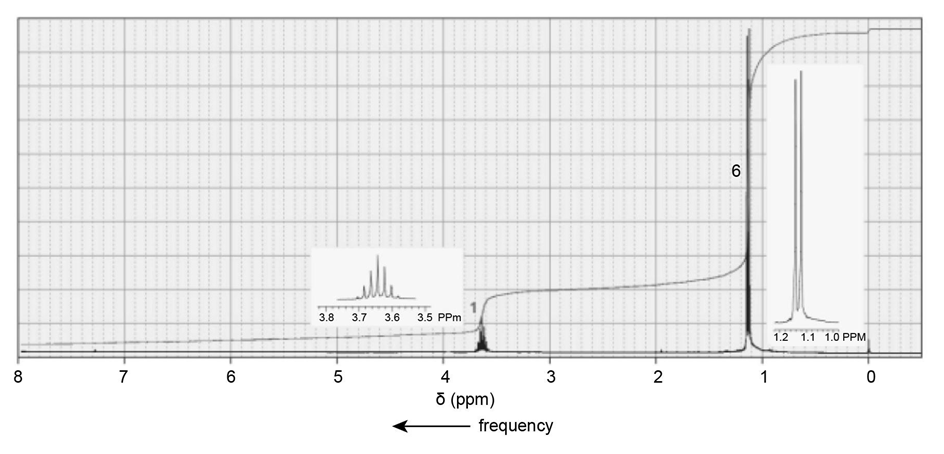
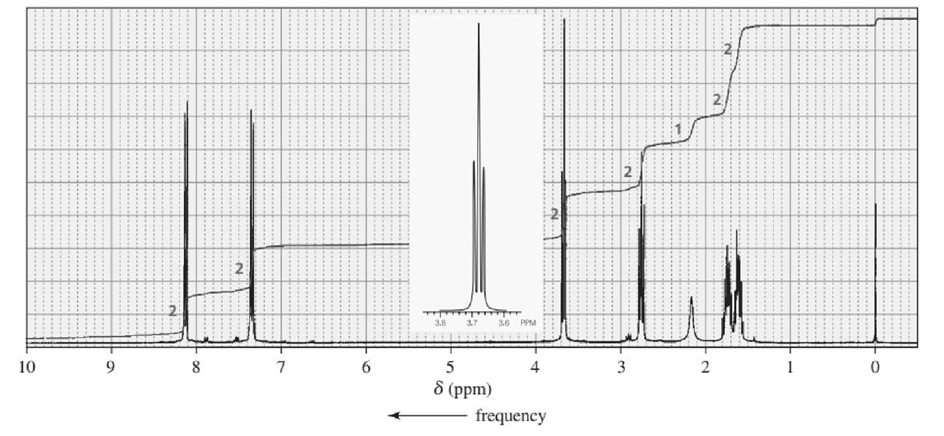
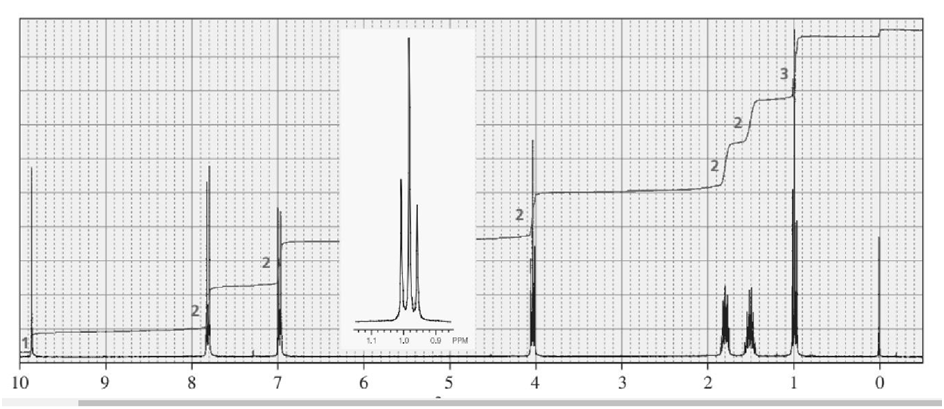
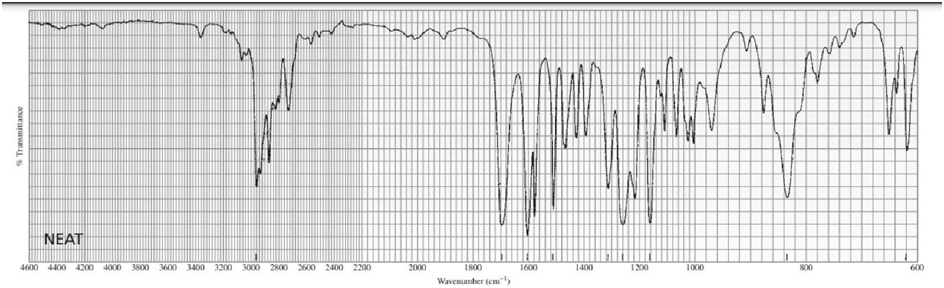
One of the spectra below is produced by 1-chloropropane and the other by 1-iodopropane. Which is which?
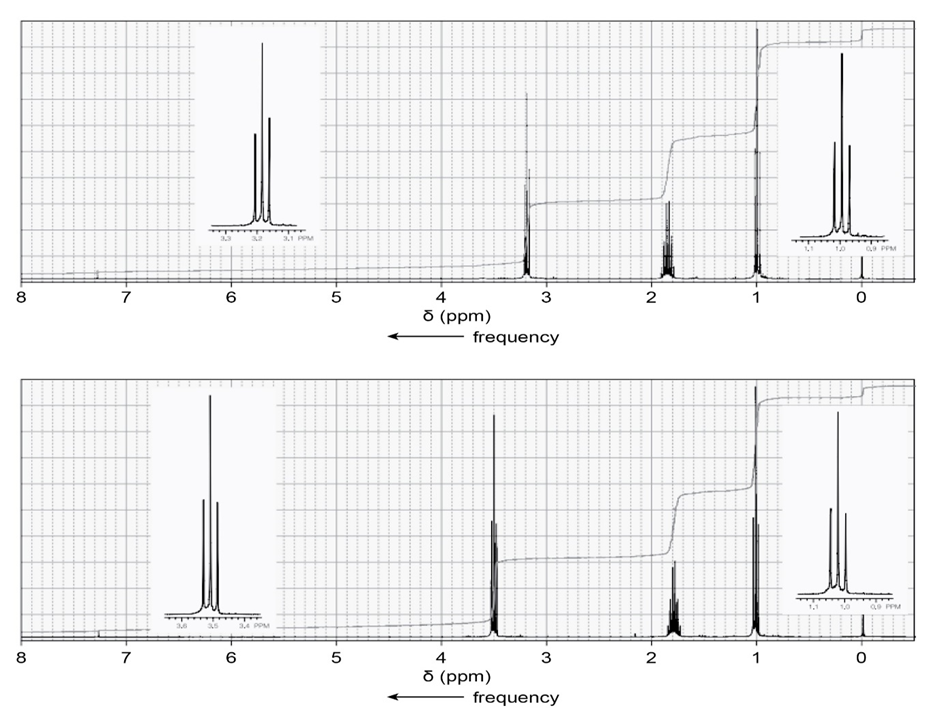
Short Answer
The signal in the range 3.3- 3.1 ppm shows the spectra produced by 1-iodopropane.
The signal in the range 3.6-3.4 ppm shows the spectra produced by 1-chloropropane.