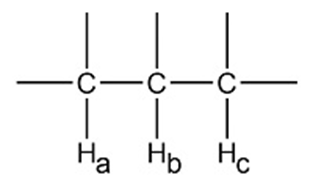
Chapter 14: Q32P (page 649)
Draw a splitting diagram for Hbwere
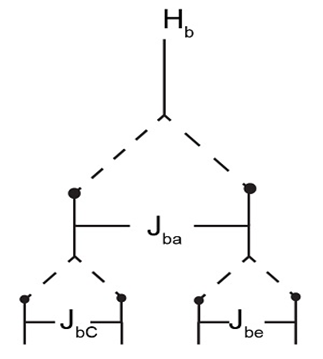
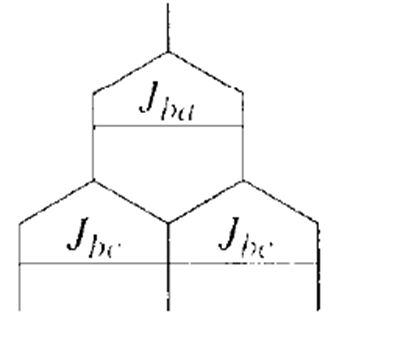
a.Jba= 12 Hz andJbc= 6Hz b.Jba= 12 Hz andJbc= 12Hz
Short Answer
The splitting of the Hb proton is shown below.
a.
b.
Learning Materials
Features
Discover
Chapter 14: Q32P (page 649)
Draw a splitting diagram for Hbwere
a.Jba= 12 Hz andJbc= 6Hz b.Jba= 12 Hz andJbc= 12Hz

The splitting of the Hb proton is shown below.
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Identify each of the following compounds from its molecular formula and its NMR spectrum:
a.
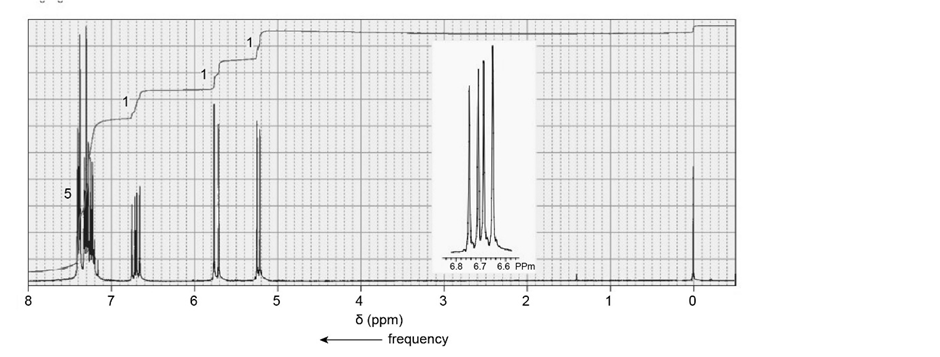
b.
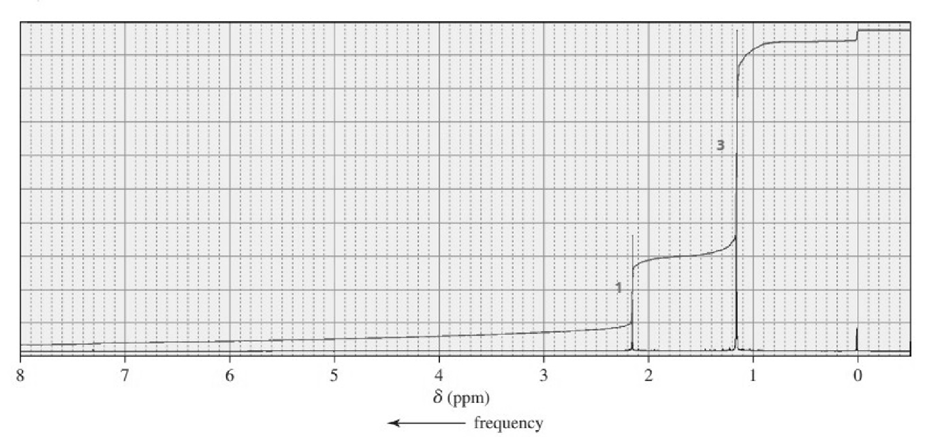
c.
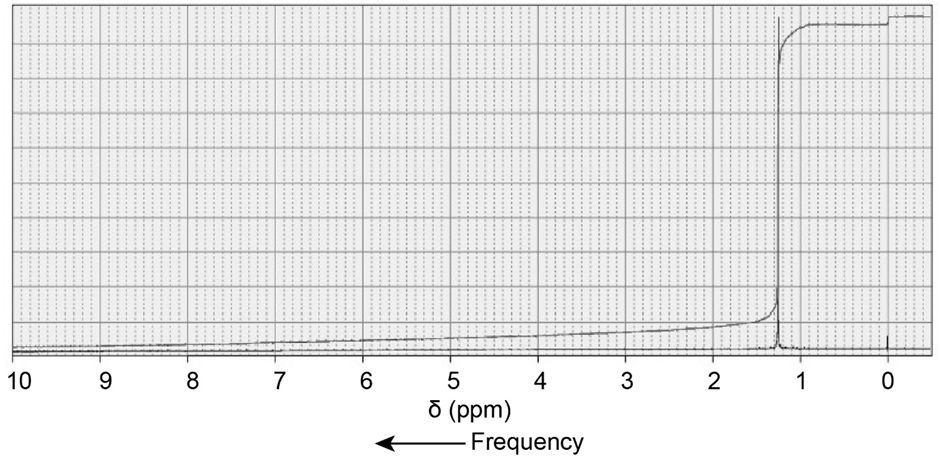
d.
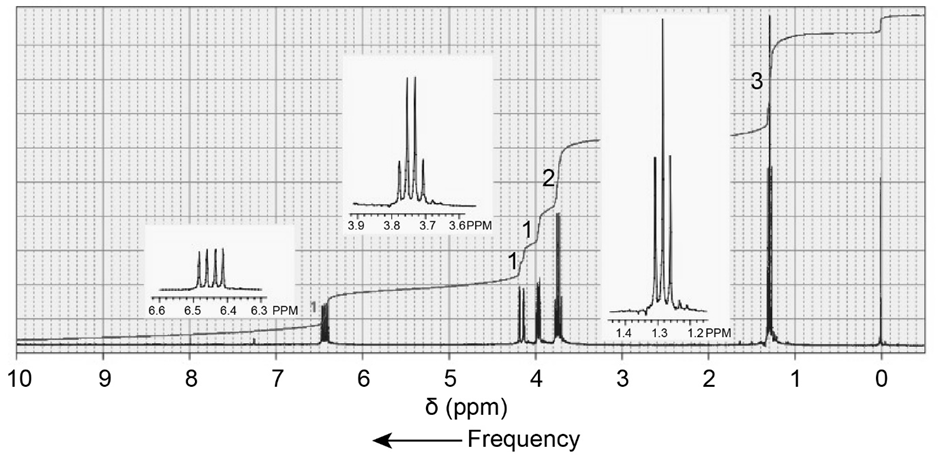
Dr. N. M. Arr was called in to help analyze the 1 H NMR spectrum of a mixture of compounds known to contain only C, H, and Br. The mixture showed two singlets-one at 1.8 ppm and the other at 2.7 ppm-with relative integrals of 1: 6, respectively. Dr. Arr determined that the spectrum was that of a mixture of bromomethane and 2-Bromo-2-methylpropane. What was the ratio of bromomethane to 2-Bromo-2-methylpropane in the mixture?
[18]-Annulene shows two signals in its 1 H NMR spectrum: one at 9.25 ppm and the other to the right of the TMS signal at -2.88 ppm. What hydrogens are responsible for each of the signals?
Identify each of the following compounds from its molecular formula and its IR and 1H NMR spectra:
a.C5H12O
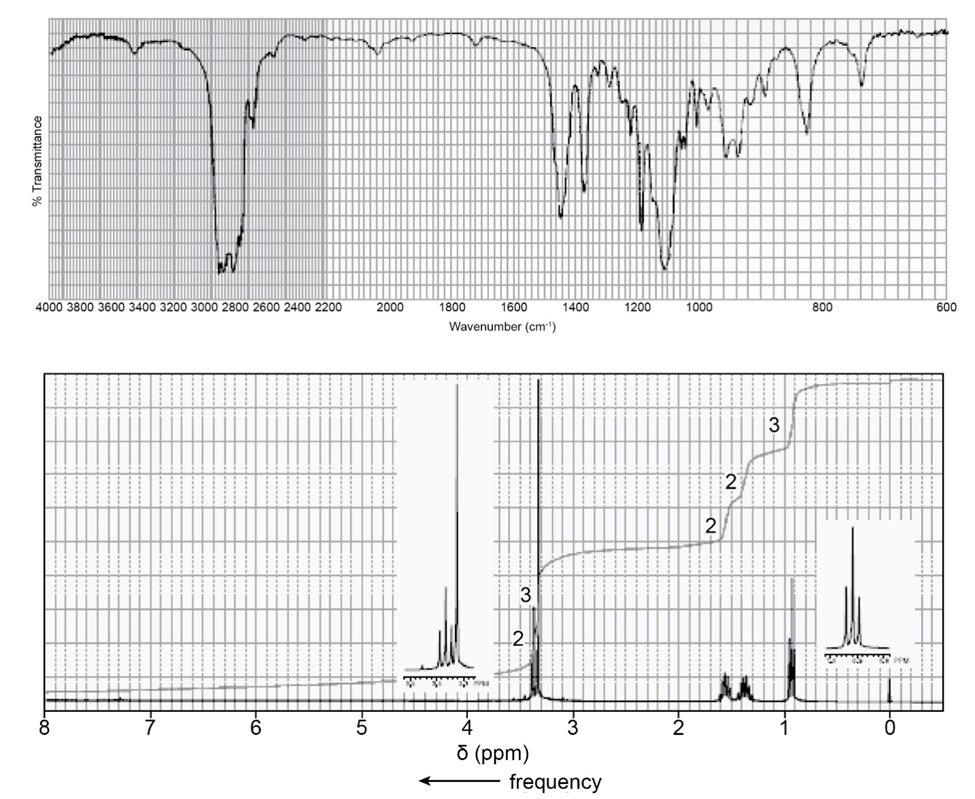
b. C6H12O2
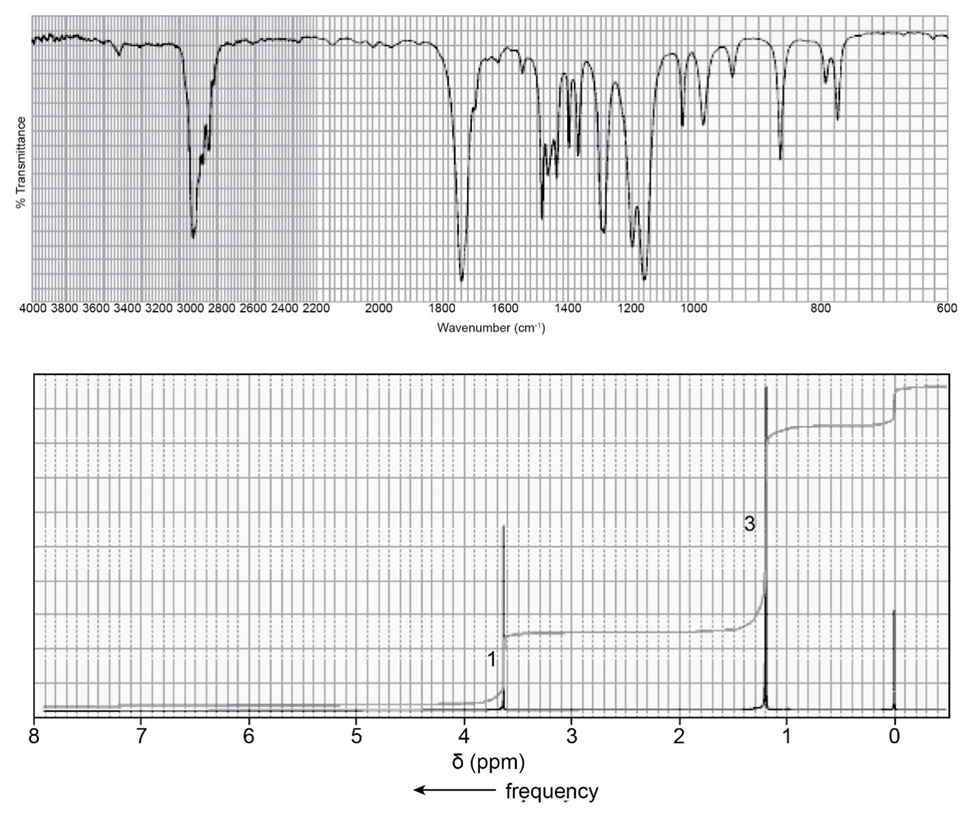
c. C4H7ClO2
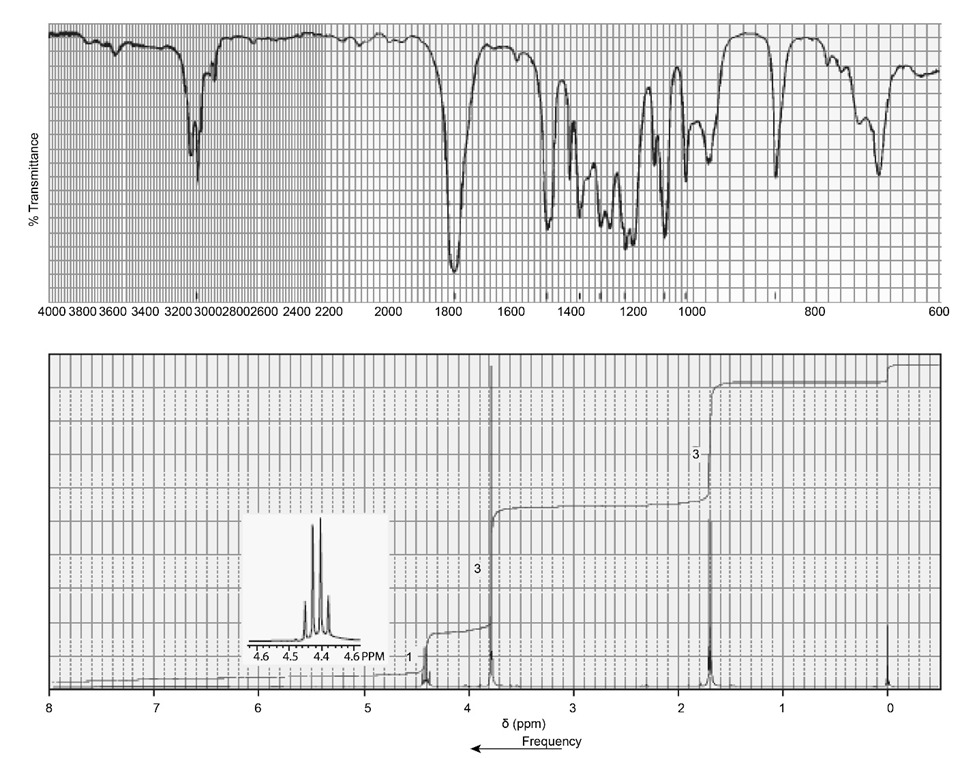
d. C4H8O2
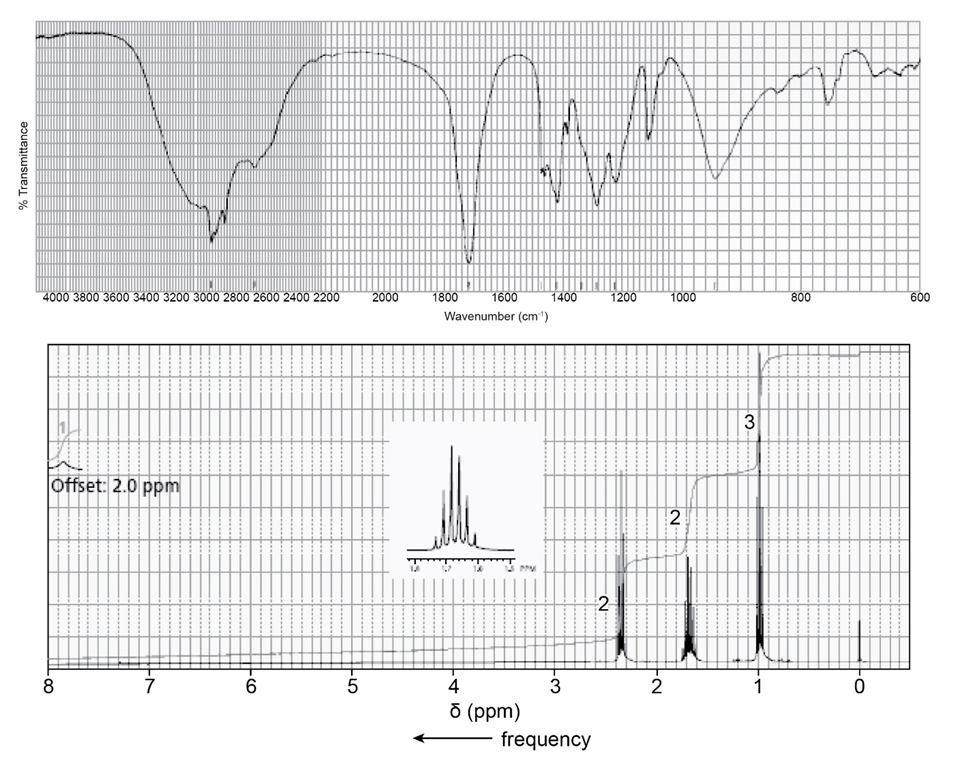
Draw a diagram like the one shown in Figure 14.12 to predict;
a. the relative intensities of the peaks in a triplet.
b. the relative intensities of the peaks in a quintet.
What do you think about this solution?
We value your feedback to improve our textbook solutions.