Chapter 14: Q14-66P (page 677)
How can the signals in the 6.5 to 6.8 ppm region of their 1H NMR spectra distinguish the following compounds?
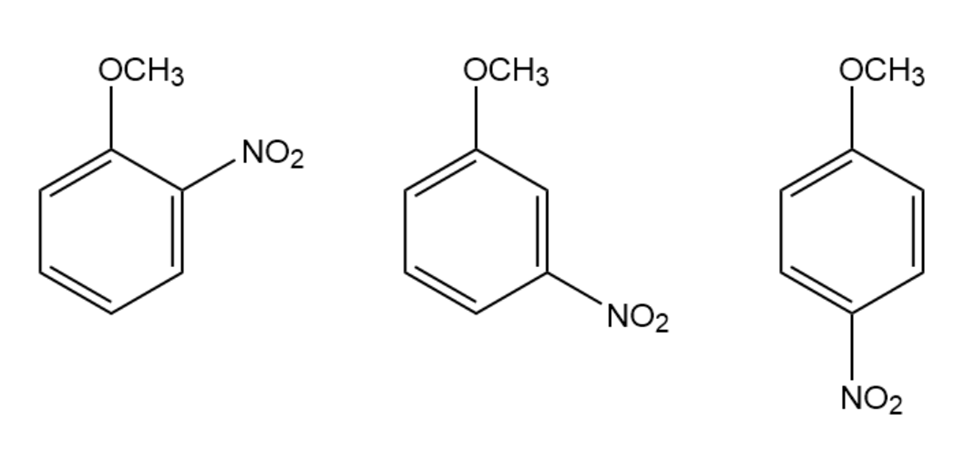
Short Answer
The following compounds can be distinguished based on the number of signal and peak type.
Learning Materials
Features
Discover
Chapter 14: Q14-66P (page 677)
How can the signals in the 6.5 to 6.8 ppm region of their 1H NMR spectra distinguish the following compounds?

The following compounds can be distinguished based on the number of signal and peak type.
All the tools & learning materials you need for study success - in one app.
Get started for free
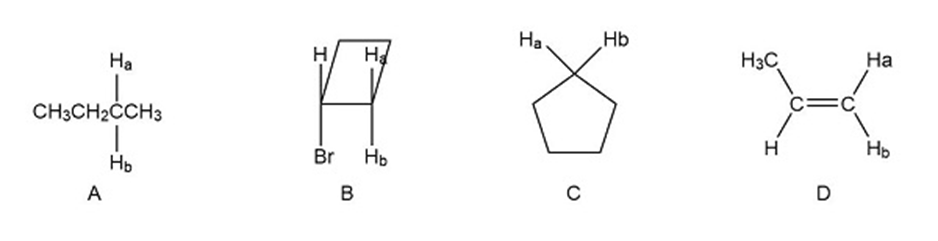
a) For the following compounds which pairs of hydrogen (Ha and Hb) are enantiotopic hydrogens?
b) which pairs are diastereotopic hydrogens?
[18]-Annulene shows two signals in its 1 H NMR spectrum: one at 9.25 ppm and the other to the right of the TMS signal at -2.88 ppm. What hydrogens are responsible for each of the signals?
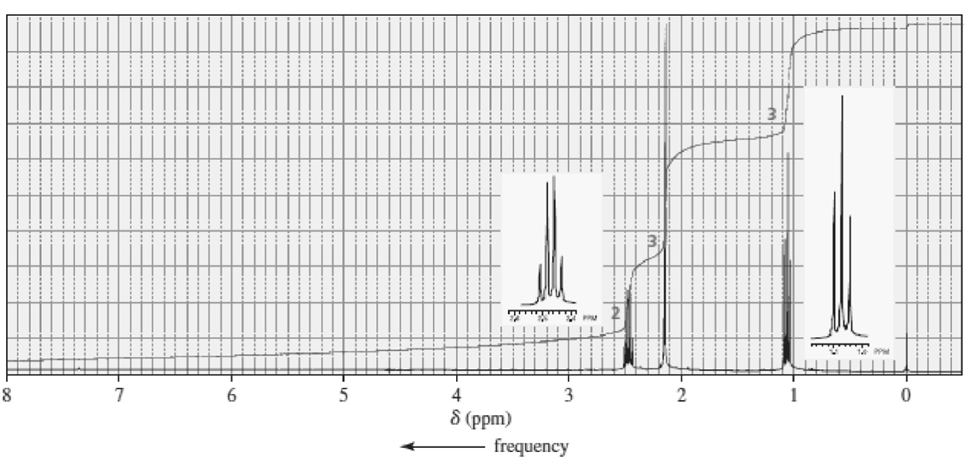
Label each set of chemically equivalent protons, using ‘a’ for the set that will be at the lowest frequency in the NMR spectrum, ‘b’ for the next lowest, and so on. Indicate the multiplicity of each signal.
a.
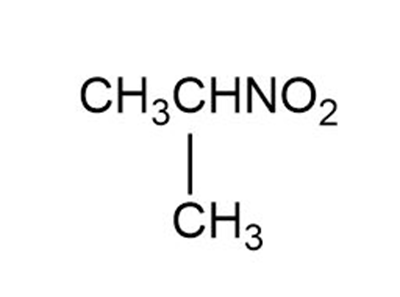
b.
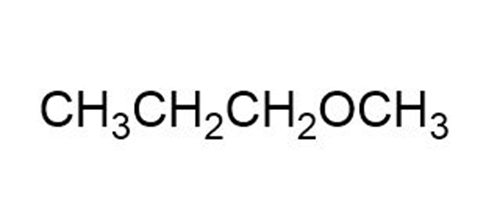
c.
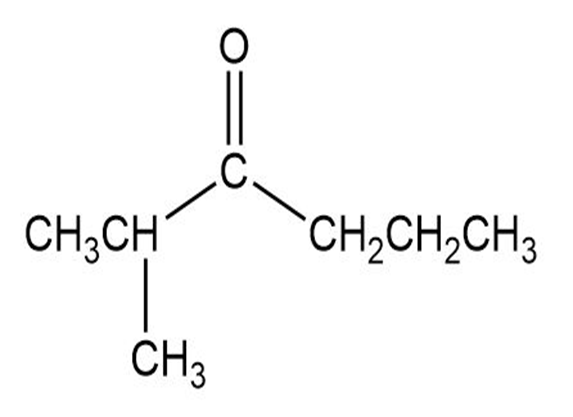
d.
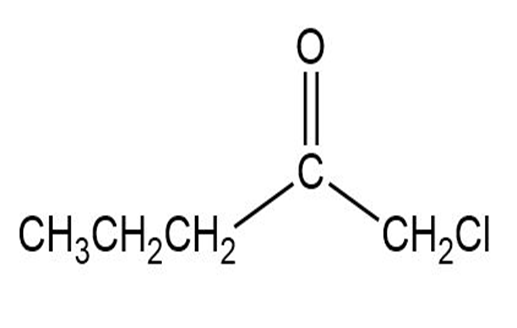
e.
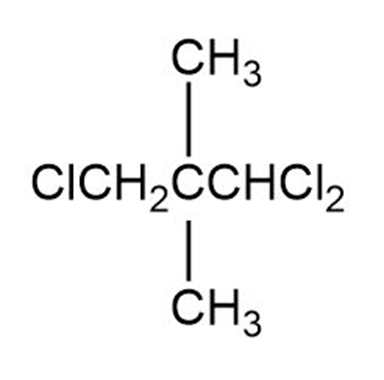
f.
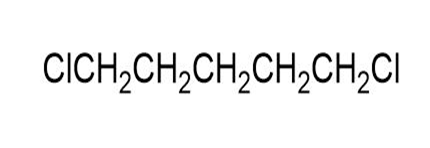
Match each of the 1HNMR spectra with one of the following compounds:
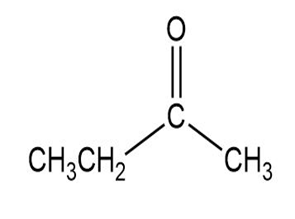
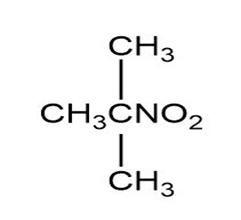
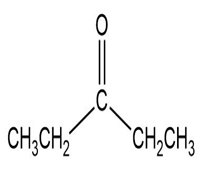
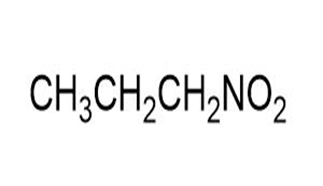
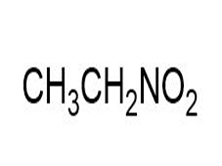
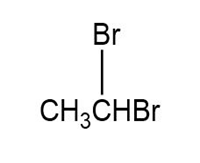
a.
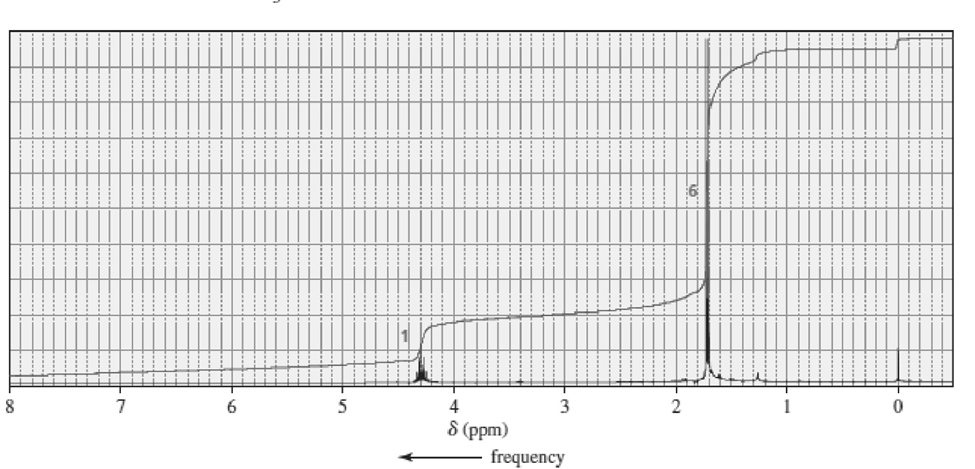
b.
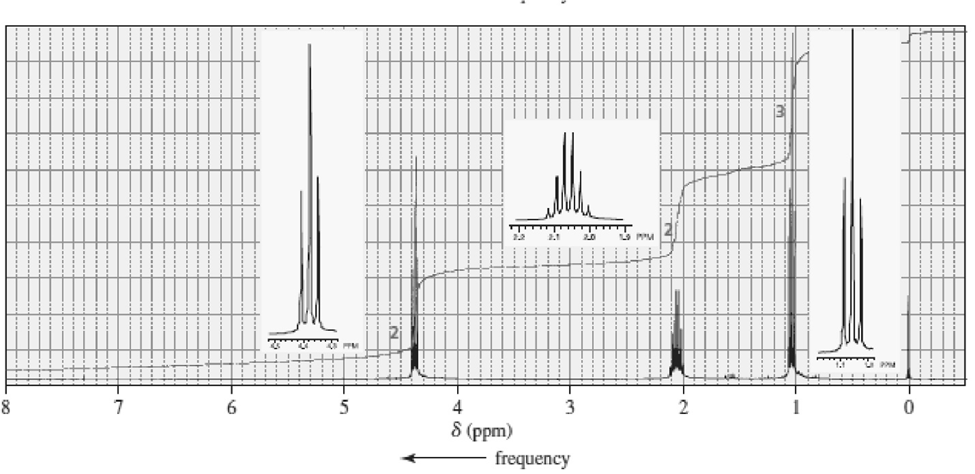
c.
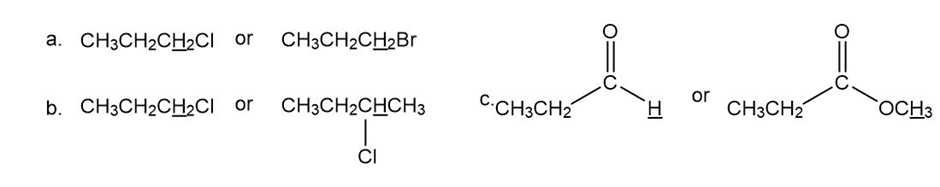
Which underlined proton (or sets of protons) has the greater chemical shift (that is, the higher frequency signal)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.