Chapter 13: Q54P (page 612)
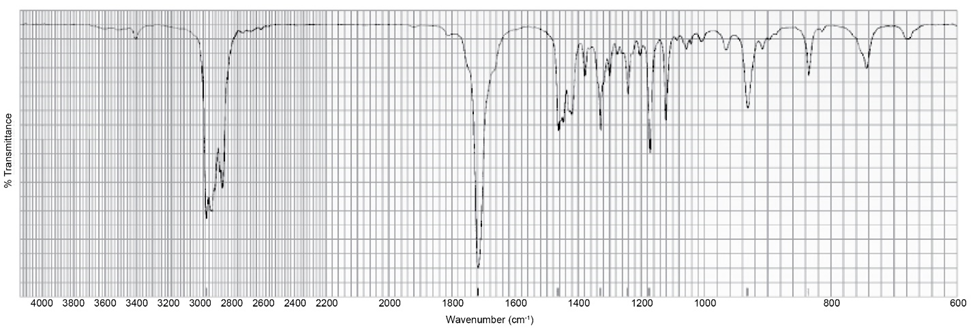
The IR spectrum of a compound with molecular formula is shown below. Identify the compound.
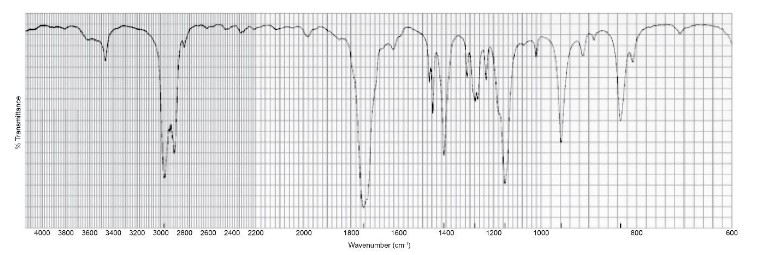
Short Answer
The compound can be:
Learning Materials
Features
Discover
Chapter 13: Q54P (page 612)
The IR spectrum of a compound with molecular formula is shown below. Identify the compound.

The compound can be:

All the tools & learning materials you need for study success - in one app.
Get started for free
Each of the IR spectra shown below is accompanied by a set of four compounds. In each case, indicate which of the four compounds is responsible for the spectrum.
(a)
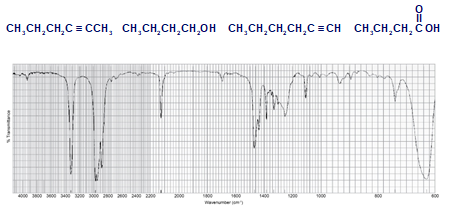
(b)
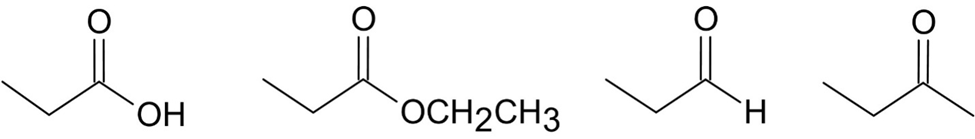
(c)
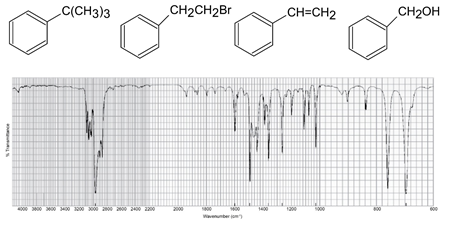
A solution of a compound in ethanol shows an absorbance of 0.52 at 236 nm in a cell with a 1 cm light path. Its molar absorptivity in ethanol at that wavelength is 12,600 M-1 cm-1. What is the concentration of the compound?
Draw structures for a saturated hydrocarbon that has a molecular ion with an m/zvalue of 128.
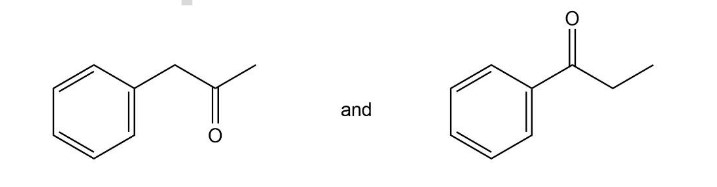
Question:How can IR spectroscopy be used to distinguish between the following compounds?
A compound gives a mass spectrum with essentially only three peaks at m/z = 77 (40%), 112 (100%), and 114 (33%). Identify the compound
What do you think about this solution?
We value your feedback to improve our textbook solutions.