Chapter 13: Q55P (page 612)
Rank the following compounds from highest wavenumber to lowest wavenumber for their C=O absorption band:
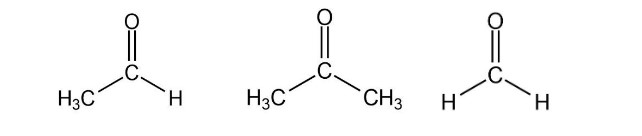
Short Answer
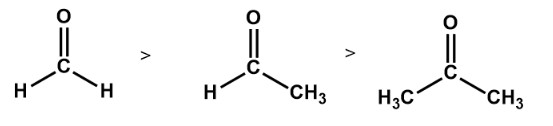
The order followed as:
Learning Materials
Features
Discover
Chapter 13: Q55P (page 612)
Rank the following compounds from highest wavenumber to lowest wavenumber for their C=O absorption band:

The order followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
How could IR spectroscopy distinguish between 1,5-hexadiene and 2,4-hexadiene?
The absorbance of a solution of a weak acid was measured under the same conditions at a series of pH values. Its conjugate base is the only species in the solution that absorbs UV light at the wavelength used. Estimate the of the acid from the data obtained.

A solution of a compound in ethanol shows an absorbance of 0.52 at 236 nm in a cell with a 1 cm light path. Its molar absorptivity in ethanol at that wavelength is 12,600 M-1 cm-1. What is the concentration of the compound?
Question: a. An oxygen containing compound shows absorption band at1700cm-1and no absorption bands at -3300cm-1, -2700cm-1 or -1100cm-1. what class of compound is it?
b. A nitrogen containing compound shows no absorption at -3400cm-1 and no absorption bands between -1700cm-1 and -1600cm-1 or between 2260cm-1 and 2220cm-1. What class of compounds is it?
What do you think about this solution?
We value your feedback to improve our textbook solutions.