Chapter 13: Q43P (page 611)
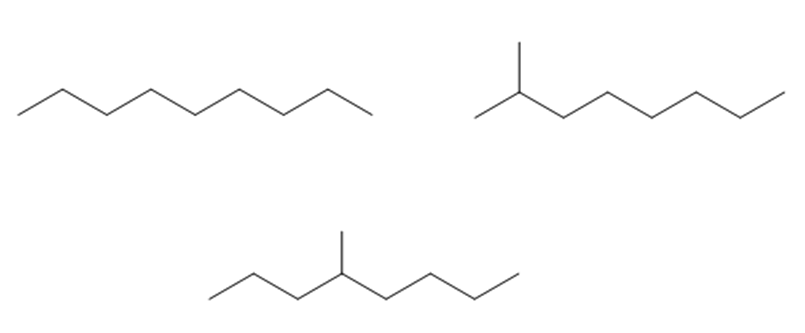
Draw structures for a saturated hydrocarbon that has a molecular ion with an m/zvalue of 128.
Short Answer
The first compound is nonane, second compound is 2-methyloctane and the third compound is 4-methyloctane.
Learning Materials
Features
Discover
Chapter 13: Q43P (page 611)
Draw structures for a saturated hydrocarbon that has a molecular ion with an m/zvalue of 128.
The first compound is nonane, second compound is 2-methyloctane and the third compound is 4-methyloctane.

All the tools & learning materials you need for study success - in one app.
Get started for free
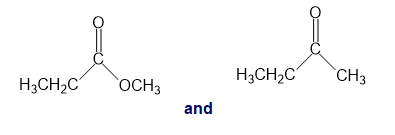
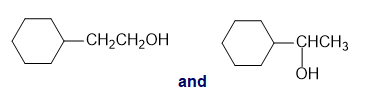
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them:
a.
b.
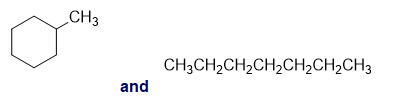
c.

d.
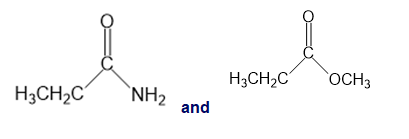
e.
f.cis-2-butene and trans-2-butene
g.
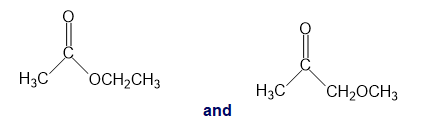
h.
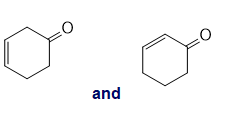
i.

j.
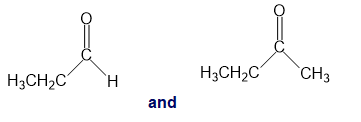
k.
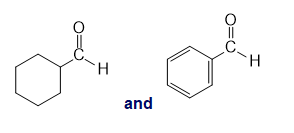
l.
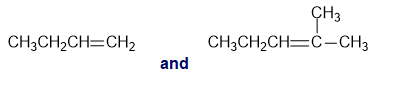
Which occurs at a larger wavenumber:
a. the C=O stretch of phenol or the C-O stretch of cyclohexanol?
b. the C=O stretch of a ketone or the C=O stretch of an amide?
c. the CN stretch of cyclohexylamine or the CN stretch of aniline?
How could IR spectroscopy distinguish between 1,5-hexadiene and 2,4-hexadiene?
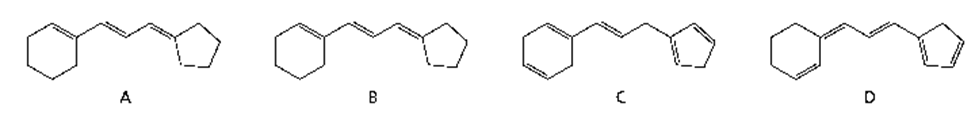
Rank the following compounds in order of increasing
Question:How can IR spectroscopy be used to distinguish between the following compounds?
What do you think about this solution?
We value your feedback to improve our textbook solutions.