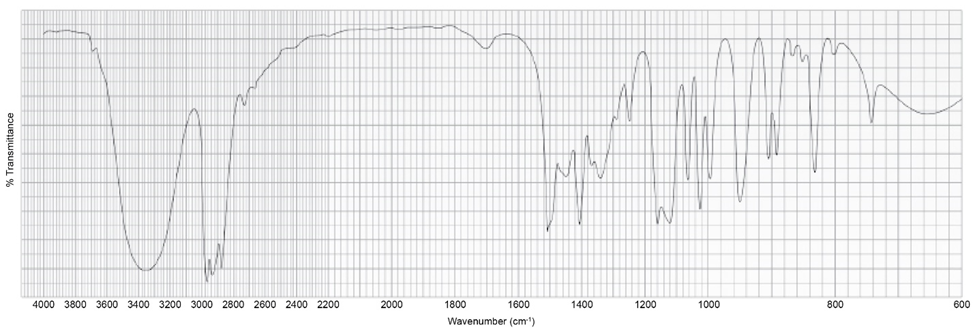
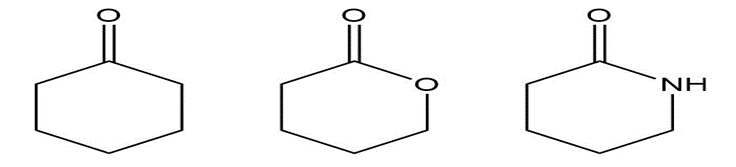
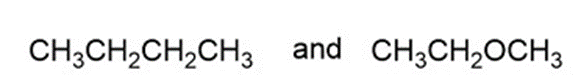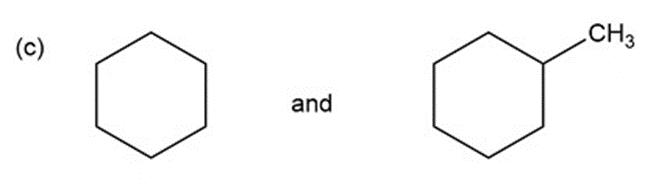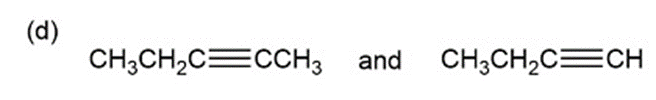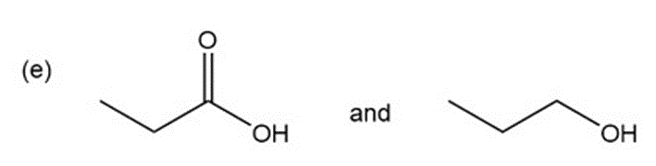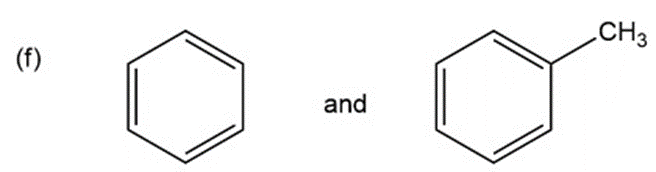Chapter 13: Q45P (page 611)
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them:
a.
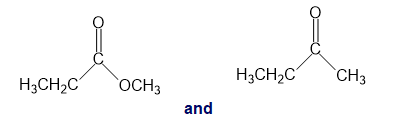
b.
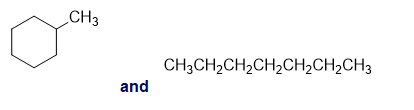
c.

d.
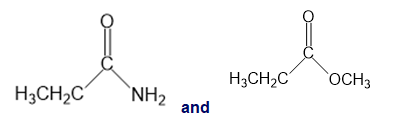
e.
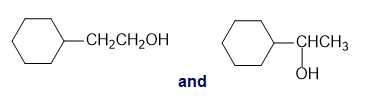
f.cis-2-butene and trans-2-butene
g.
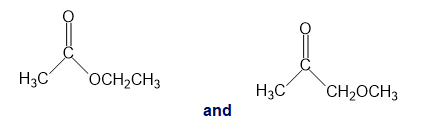
h.
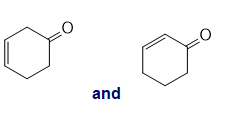
i.

j.
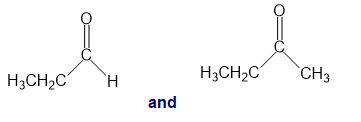
k.
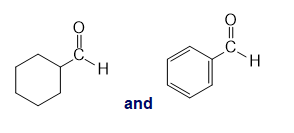
l.
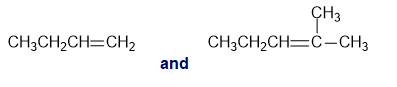
Short Answer
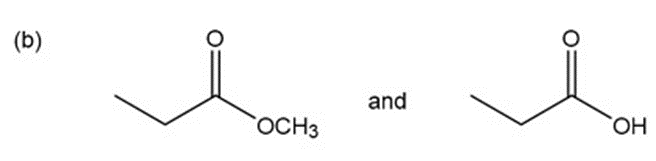
a.An absorption band at is present for the ester.
b.An absorption band at is present for the second compound.
c.An absorption band at 3650 to is present for the first compound.
d.An absorption band at 3500 to is present for the first compound.
e.The second compound has an absorption of 1385 to .
f.The trans isomer has an absorption in the range of 980 to role="math" localid="1652421519275" . The cis isomer has an absorption around 730 to role="math" localid="1652421536620" .
g. The C=O absorption band possess a value of role="math" localid="1652421551920" for the first compound. The ketone which is the second compound possess a wave number of role="math" localid="1652421559908" .
h. The C=O absorption band comprise a larger wavenumber for role="math" localid="1652421619465" ,role="math" localid="1652421651997" -unsaturated ketone and the value is role="math" localid="1652421662734" . The role="math" localid="1652421671530" unsaturated ketone has a value ofrole="math" localid="1652421680991" .
i. The alkene has an absorption band at 1680 to role="math" localid="1652421694246" due to the C=C stretching vibration and at 3100 to role="math" localid="1652421704176" due to anrole="math" localid="1652421712844" C-H stretching vibration. The alkyne has an absorption band at 2260 to .
j.An absorption band at 2820 and due to aldehyde C-H stretching vibration is present for the first compound.
k.An absorption band at and (aromatic ring stretching vibrations) and at 3100 to stretching vibration is present for the compound with benzene ring and absent for the compound with cyclohexane ring.
l. Absorption band at and due to an C-H bending vibration that is present for terminal alkene.