Chapter 13: Q65P (page 616)
Question: Draw a structure for a carboxylic acid that has a molecular ion with an m/zvalue of 116.
Short Answer
Answer
The possible structures are given below.
Learning Materials
Features
Discover
Chapter 13: Q65P (page 616)
Question: Draw a structure for a carboxylic acid that has a molecular ion with an m/zvalue of 116.
Answer
The possible structures are given below.
All the tools & learning materials you need for study success - in one app.
Get started for free
Rank the following compounds from highest wavenumber to lowest wavenumber for their C¬O absorption band:
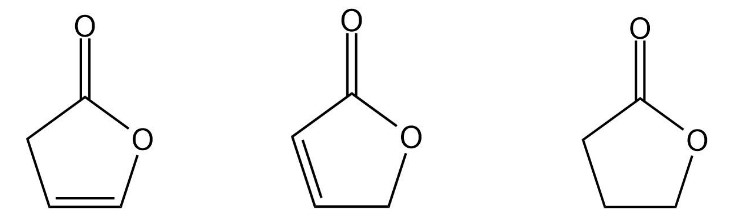
What hydrocarbons that contain a six-membered ring will have a molecular ion peak at m/z = 112?
Question: which of the following compounds has a vibration that is infrared inactive.
1-butyne, 2-butyne, H2, H2O, Cl2 and ethene
In the following boxes, list the types of bonds and the approximate wavenumber at which each type of bond is expected to show an IR absorption:
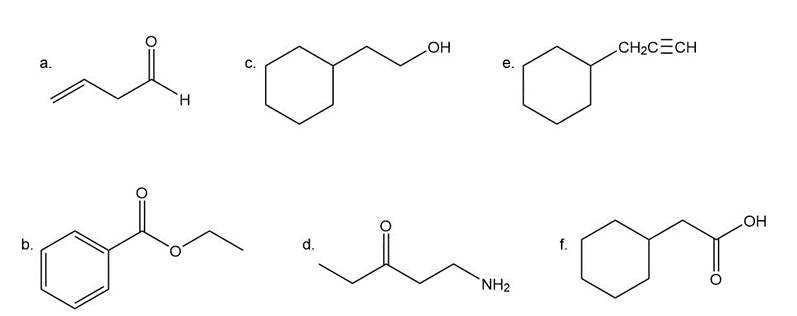
Give approximate wavenumbers for the major characteristic IR absorption bands that would be given by each of the following compounds:
What do you think about this solution?
We value your feedback to improve our textbook solutions.