Chapter 13: 31P (page 599)
Question: which of the following compounds has a vibration that is infrared inactive.
1-butyne, 2-butyne, H2, H2O, Cl2 and ethene
Short Answer
Answer
The compounds that are infrared active are 2-butyne, H2, Cl2 and ethene
Learning Materials
Features
Discover
Chapter 13: 31P (page 599)
Question: which of the following compounds has a vibration that is infrared inactive.
1-butyne, 2-butyne, H2, H2O, Cl2 and ethene
Answer
The compounds that are infrared active are 2-butyne, H2, Cl2 and ethene
All the tools & learning materials you need for study success - in one app.
Get started for free
How could IR spectroscopy distinguish between 1,5-hexadiene and 2,4-hexadiene?
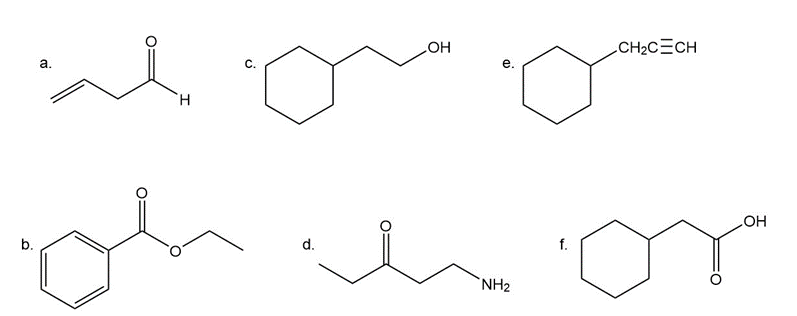
Give approximate wavenumbers for the major characteristic IR absorption bands that would be given by each of the following compounds:
Which shows an O-H stretch at a larger wavenumber: ethanol dissolved in carbon disulfide or an undiluted sample of ethanol?
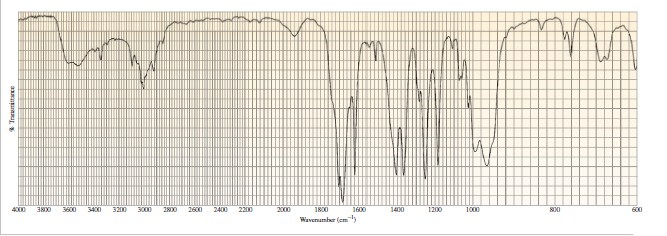
A compound with molecular formula C4H6O gives the infrared spectrum shown here. Identify the compound.
Question: for each of the following pairs of compounds, name one absorption band that can be used to distinguish between them.
a.
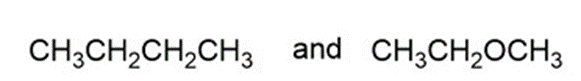
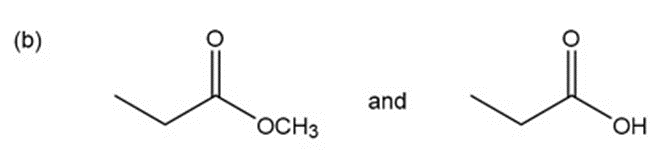
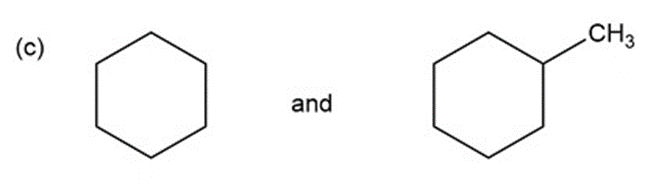
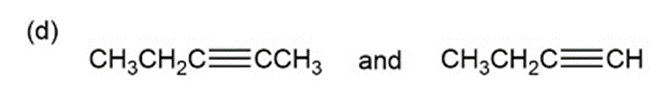
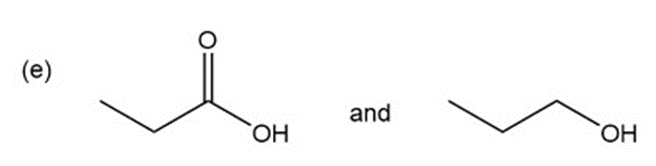
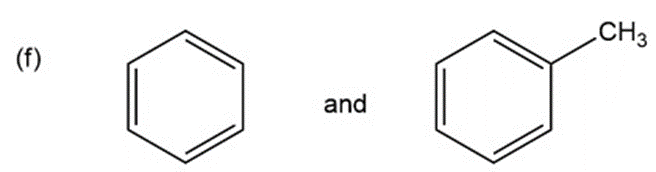
What do you think about this solution?
We value your feedback to improve our textbook solutions.