Chapter 13: 13-33P (page 602)
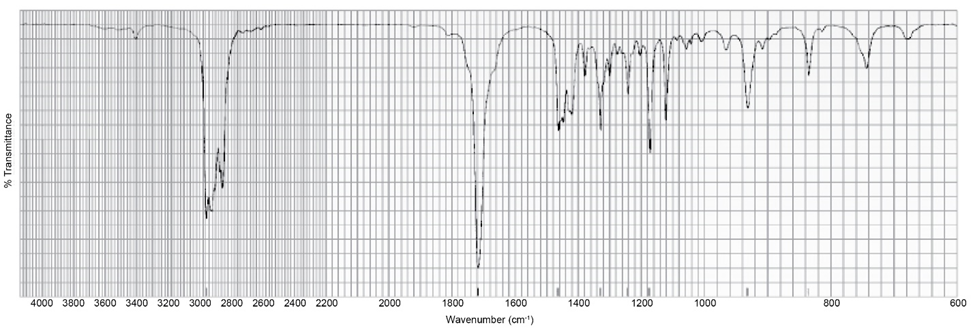
A compound with molecular formula C4H6O gives the infrared spectrum shown here. Identify the compound.
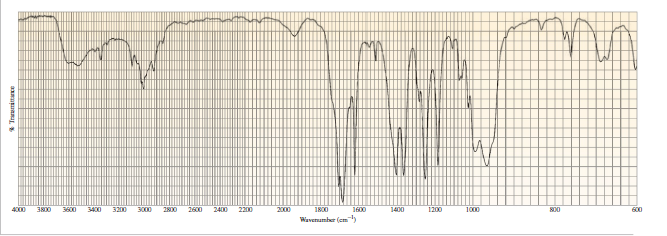
Short Answer
The compound here is methyl vinyl ketone i.e., but-3-en-2-one.
Learning Materials
Features
Discover
Chapter 13: 13-33P (page 602)
A compound with molecular formula C4H6O gives the infrared spectrum shown here. Identify the compound.

The compound here is methyl vinyl ketone i.e., but-3-en-2-one.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Rank the following compounds from highest wavenumber to lowest wavenumber for their C=O absorption bands:
a.
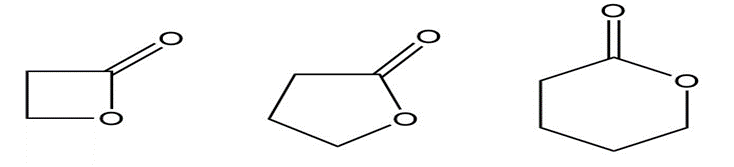
b.
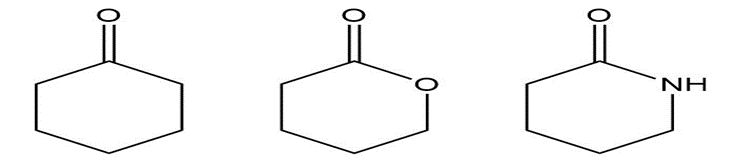
Why is the C-O absorption band of 1-hexanol at a smaller wavenumber (1060 cm-1) than the C=O absorption band of pentanoic acid (1220 cm-1)?
Each of the IR spectra shown below is the spectrum of one of the following compounds. Identify the compound that produced each spectrum.
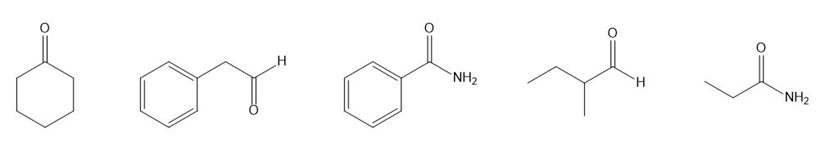
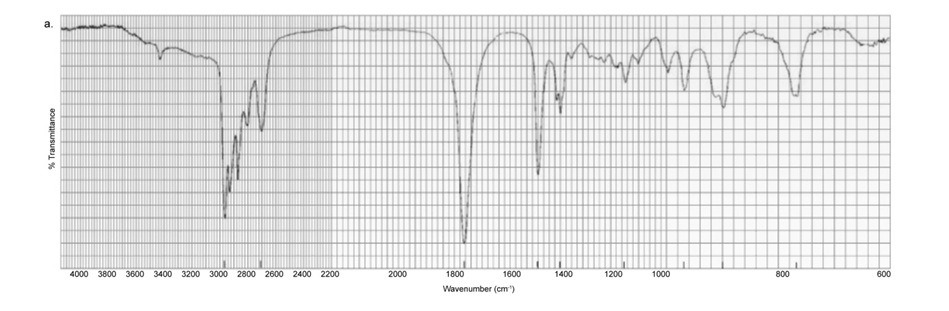
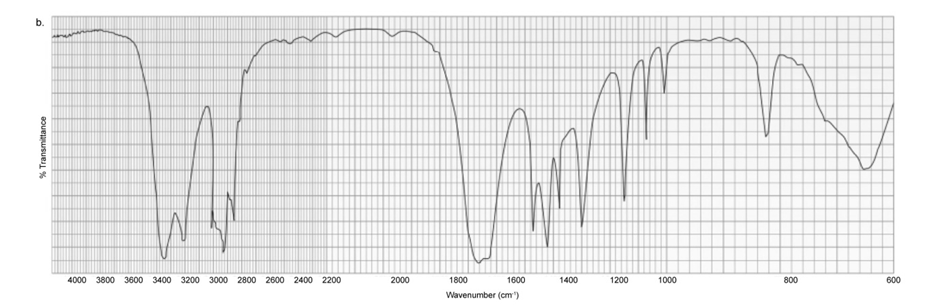
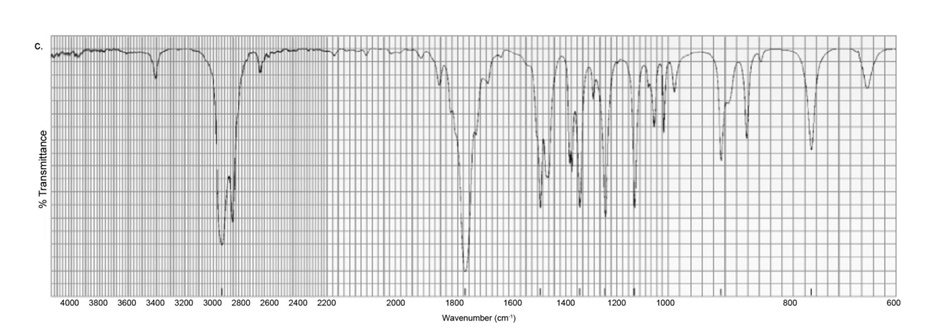
Each of the IR spectra shown below is accompanied by a set of four compounds. In each case, indicate which of the four compounds is responsible for the spectrum.
(a)
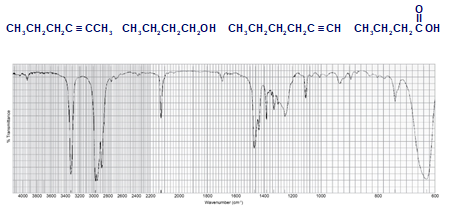
(b)
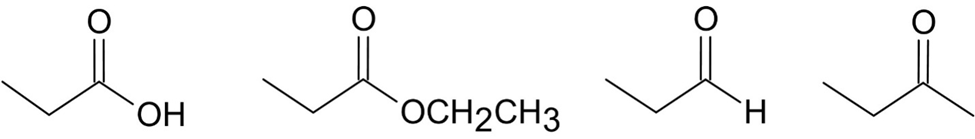
(c)
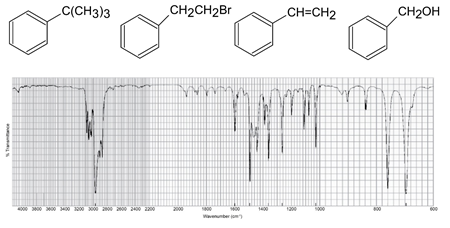
What do you think about this solution?
We value your feedback to improve our textbook solutions.