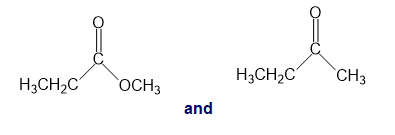
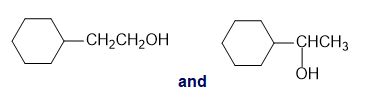
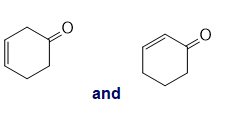
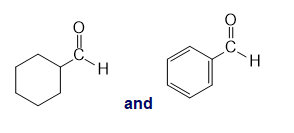
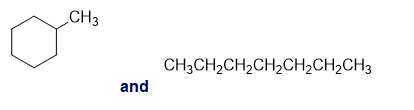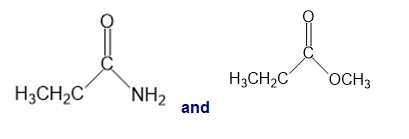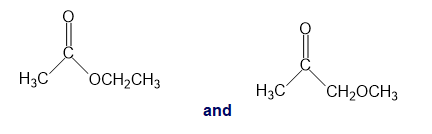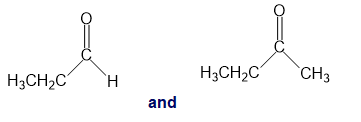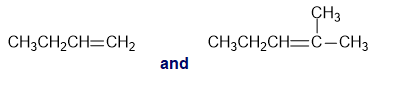Chapter 13: Q26P (page 567)
Which shows an O-H stretch at a larger wavenumber: ethanol dissolved in carbon disulfide or an undiluted sample of ethanol?
Short Answer
An undiluted sample of ethanol shows an O-H stretch at a larger wavenumber than ethanol dissolved in carbon disulfide.