Chapter 13: Q50P (page 612)
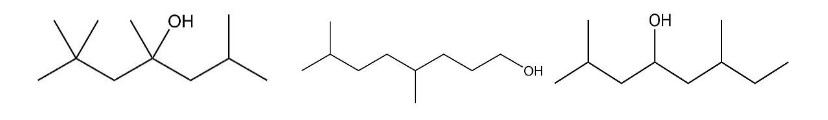
A mass spectrum shows significant peaks at m/z = 87, 115, 140, and 143. Which of the following compounds is responsible for that mass spectrum?
Short Answer
The compound is
Learning Materials
Features
Discover
Chapter 13: Q50P (page 612)
A mass spectrum shows significant peaks at m/z = 87, 115, 140, and 143. Which of the following compounds is responsible for that mass spectrum?

The compound is

All the tools & learning materials you need for study success - in one app.
Get started for free
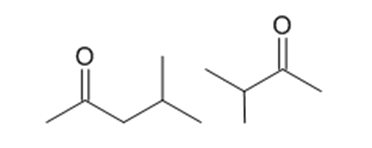
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them:
a.
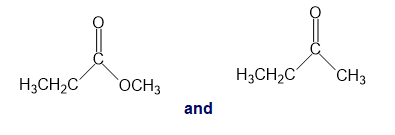
b.
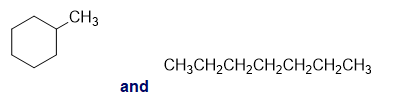
c.

d.
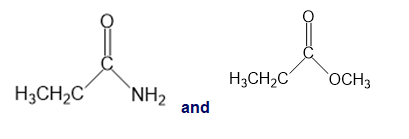
e.
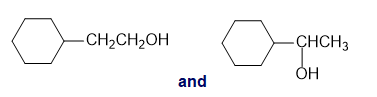
f.cis-2-butene and trans-2-butene
g.
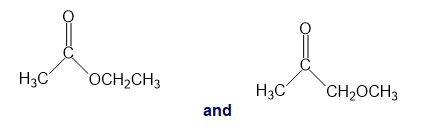
h.
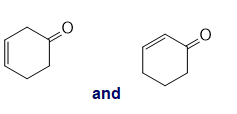
i.

j.
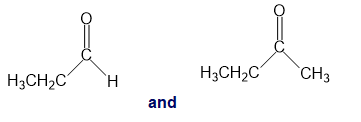
k.
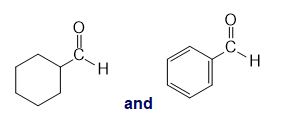
l.
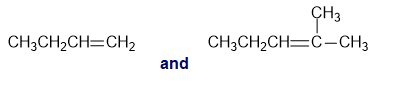
Which shows an absorption band at a larger wavenumber: a carbonyl group bonded to a sp3 carbon or a carbonyl group bonded to an sp2 carbon of an alkene?
What peaks in their mass spectra can be used to distinguish between the following compounds?
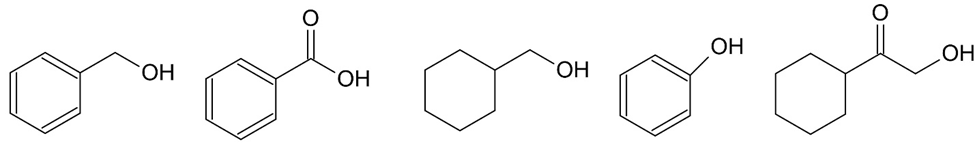
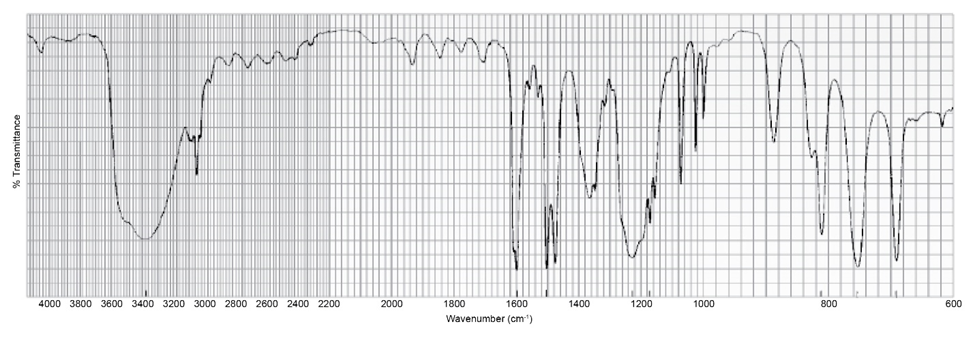
Which one of the following five compounds produced the IR spectrum shown below?
What do you think about this solution?
We value your feedback to improve our textbook solutions.