Chapter 13: Q53P (page 612)
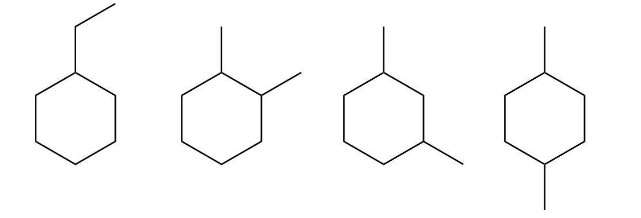
What hydrocarbons that contain a six-membered ring will have a molecular ion peak at m/z = 112?
Short Answer
Possible hydrocarbons are:
Learning Materials
Features
Discover
Chapter 13: Q53P (page 612)
What hydrocarbons that contain a six-membered ring will have a molecular ion peak at m/z = 112?
Possible hydrocarbons are:

All the tools & learning materials you need for study success - in one app.
Get started for free
A solution of a compound in hexane shows an absorbance of 0.40 at 252 nm in a cell with a 1 cm light path. What is the molar absorptivity of the compound in hexane at 252 nm?
Question: a. An oxygen containing compound shows absorption band at1700cm-1and no absorption bands at -3300cm-1, -2700cm-1 or -1100cm-1. what class of compound is it?
b. A nitrogen containing compound shows no absorption at -3400cm-1 and no absorption bands between -1700cm-1 and -1600cm-1 or between 2260cm-1 and 2220cm-1. What class of compounds is it?
Which shows an O-H stretch at a larger wavenumber: ethanol dissolved in carbon disulfide or an undiluted sample of ethanol?
How could IR spectroscopy distinguish between 1,5-hexadiene and 2,4-hexadiene?
Draw structures for a saturated hydrocarbon that has a molecular ion with an m/zvalue of 128.
What do you think about this solution?
We value your feedback to improve our textbook solutions.