Chapter 13: Q52P (page 612)
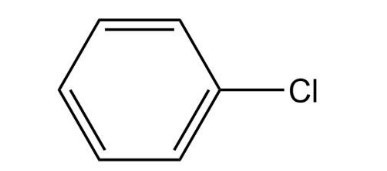
A compound gives a mass spectrum with essentially only three peaks at m/z = 77 (40%), 112 (100%), and 114 (33%). Identify the compound
Short Answer
The compound is benzoyl chloride
Learning Materials
Features
Discover
Chapter 13: Q52P (page 612)
A compound gives a mass spectrum with essentially only three peaks at m/z = 77 (40%), 112 (100%), and 114 (33%). Identify the compound
The compound is benzoyl chloride

All the tools & learning materials you need for study success - in one app.
Get started for free
Given that the force constants are similar for C¬H and C¬C bonds, explain why the stretching vibration of a C¬H bond occurs at a larger wavenumber.
Question:How do you know that the absorption band at -1700cm-1in figure 13.23 is due to a C-O bond and not a C-N bond.
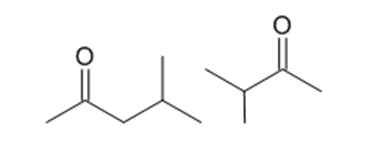
What peaks in their mass spectra can be used to distinguish between the following compounds?
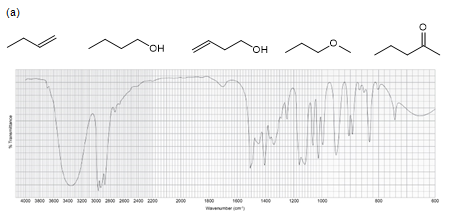
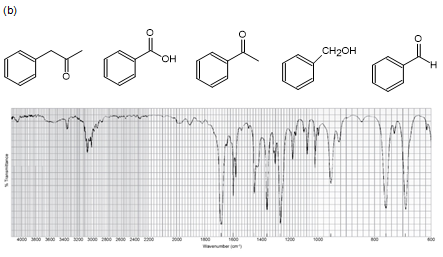
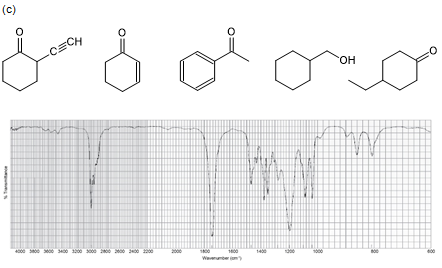
Five compounds are shown for each of the IR spectra below. Indicate which of the five compounds is responsible for each spectrum.
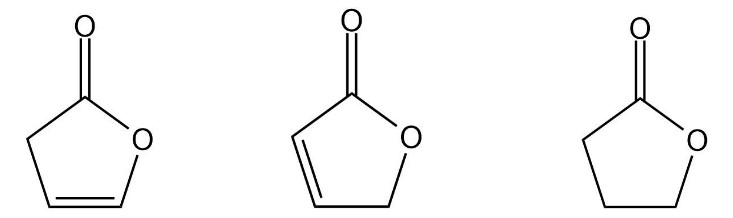
Rank the following compounds from highest wavenumber to lowest wavenumber for their C¬O absorption band:
What do you think about this solution?
We value your feedback to improve our textbook solutions.