Chapter 5: Alkenes: Structure, Nomenclature, and an Introduction to Reactivity, Thermodynamics and Kinetics
Q5-42P
Draw the condensed structure for each of the following:
a. (Z)-1,3,5-tribromo-2-pentene
b. (Z)-3-methyl-2-heptene
c. (E)-1,2-dibromo-3-isopropyl-2-hexene
d. vinyl bromide
e. 1,2-dimethylcyclopentene
f. diallyl amine
Q-5-43P
Draw the Skeletal structure for each of the following:
a. (Z)-1,3,5-tribromo-2-pentene
b. (Z)-3-methyl-2-heptene
c. (E)-1,2-dibromo-3-isopropyl-2-hexene
d. vinyl bromide
e. 1,2-dimethylcyclopentene
f. diallyl amine
Q-5-44P
a. Draw the condensed structures and give the systematic names for all the alkenes with molecular formula C6H12, ignoring stereoisomers.
(Hint:There are 13.)
b. Which of the alkenes have Eand Zisomers?
c. Which of the alkenes is the most stable?
d. Which of the alkenes is the least stable?
Q5-45P
Name the following:
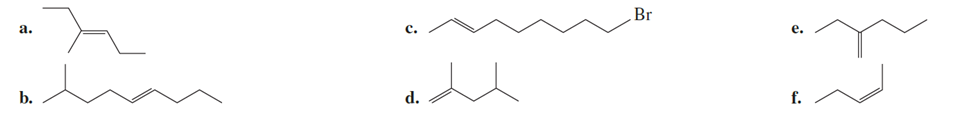
Q-5-46P
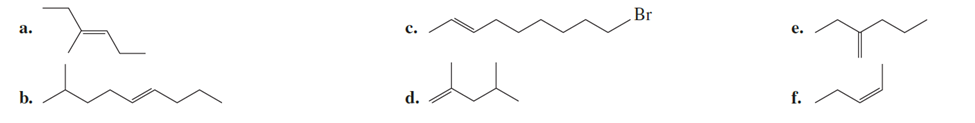
Some compounds are given below.(Question no 45)
a. Which is the most stable?
b. Which is the least stable?
Q-5-47P
a. Which is the most stable: 3,4-dimethyl-2-hexene, 2,3-dimethyl-2-hexene, or 4,5-dimethyl-2-hexene?
b. Which compound has the largest heat of hydrogenation?
c. Which compound has the smallest heat of hydrogenation?
Q5-48P
Draw curved arrows to show the flow of electrons responsible for the conversion of the reactants to products
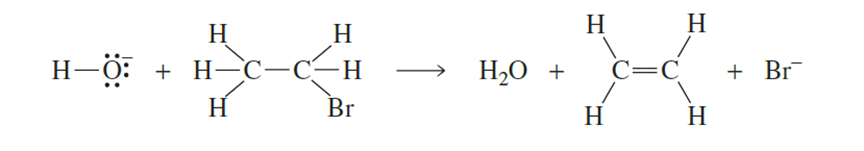
Q5-49P
Draw the skeletal structure of 3,3-dimethyl-7-(1-methyl ethyl)-6-(1-methyl propyl) decane.
Q5-50P
In a reaction in which reactant A is in equilibrium with product B at
25 °C, what relative amounts of A and B are present at equilibrium if ΔG° at 25 °C is
a. 2.72 kcal/mol?
b. 0.65 kcal/mol?
c. -2.72 kcal/mol?
d. -0.65 kcal/mol?
Q5-51P
Which bond is stronger? Briefly explain why.

