Chapter 11: Q26P (page 570)
Propose a mechanism for each reaction.(a)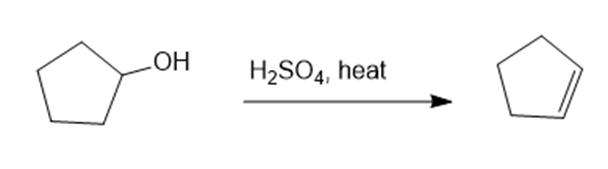
(b)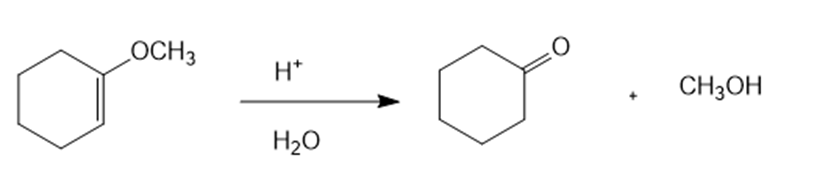
(c)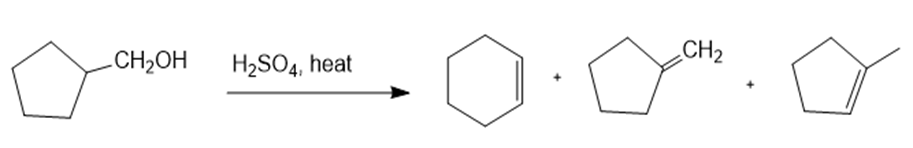
(d) 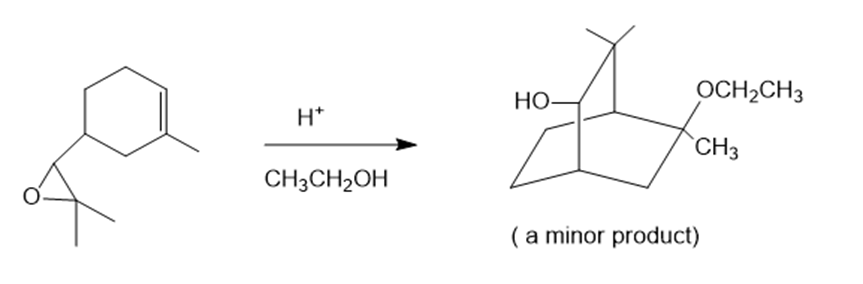
(a)
(b)
(c)
(d) 
Short Answer
(a) 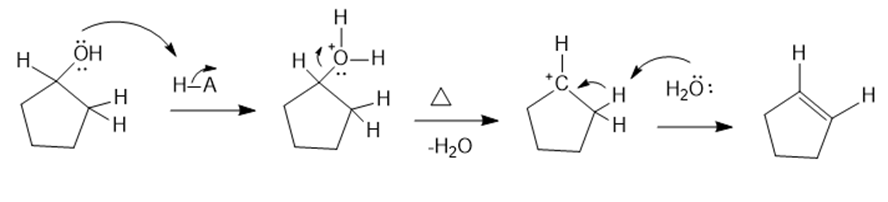
(b)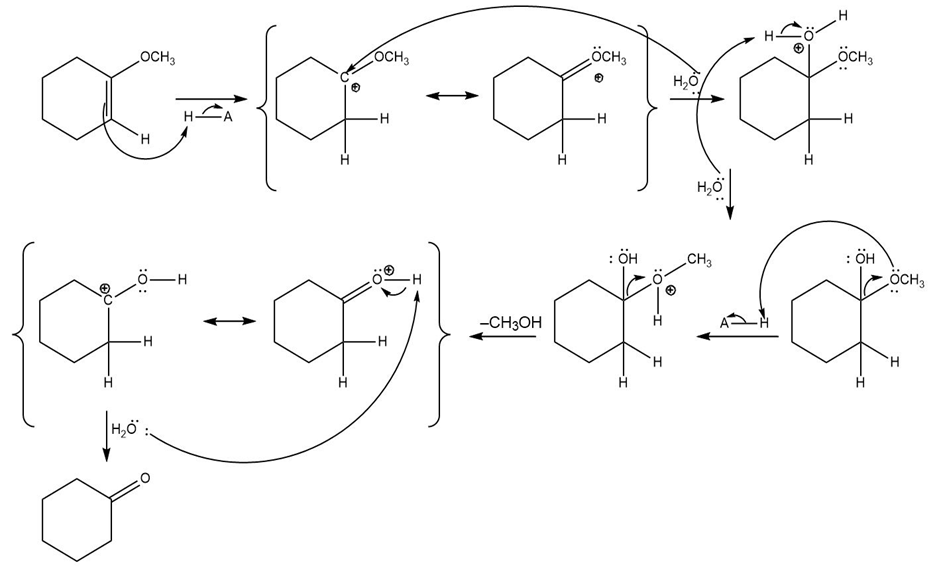
(c) 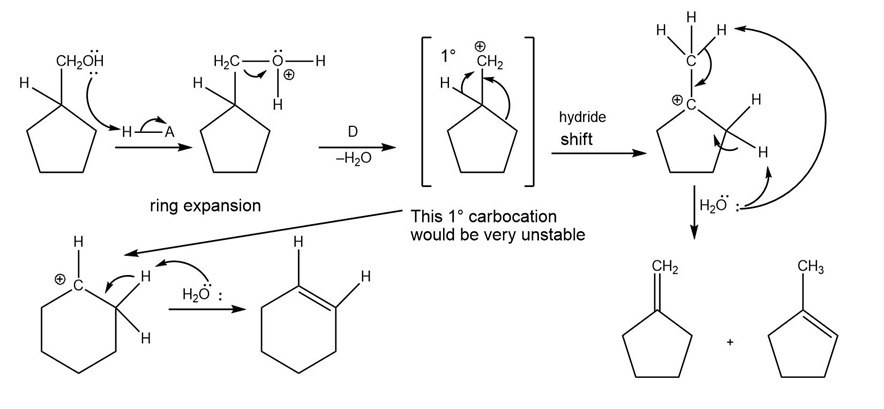
(d) 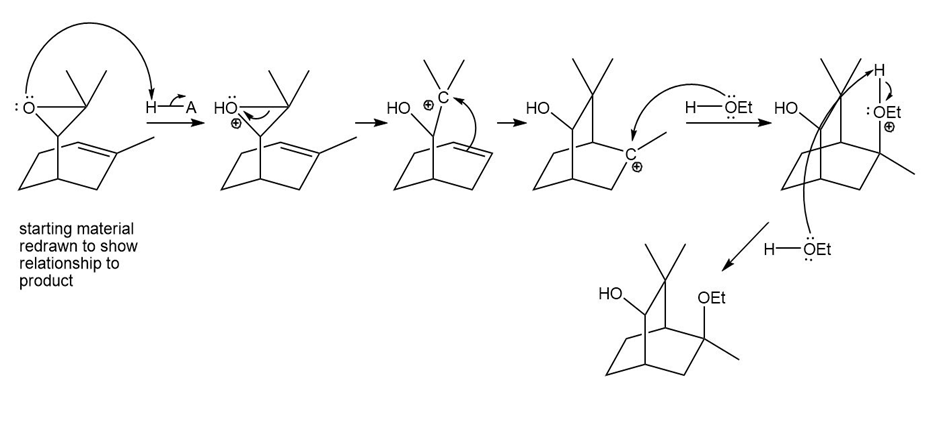
Learning Materials
Features
Discover
Chapter 11: Q26P (page 570)
(a)
(b)
(c)
(d) 
(a) 
(b)
(c) 
(d) 
All the tools & learning materials you need for study success - in one app.
Get started for free
To practice working through the early parts of a multistep synthesis, devise syntheses of
(a) pentan-3-one from alcohols containing no more than three carbon atoms.
(b) 3-ethylpentan-2-one from compounds containing no more than three carbon atoms.
Predict the products of the following reactions.
(a) cyclohexylmethanol + TsCl / pyridine (b) product of (a) + LiAlH4
(c) 1- methylcyclohexanol + H2SO4, heat (d) product of (c) + H2, Pt
Use resonance forms of the conjugate bases to explain why methanesulfonic acid (CH3SO3H,pKa= -2.6) is a much stronger acid than acetic acid (CH3COOH,pKa =4.8)
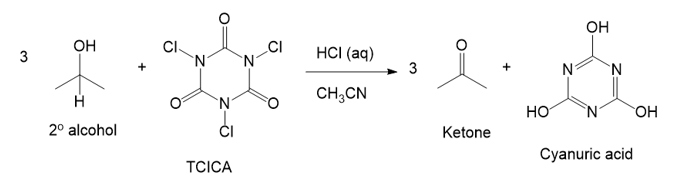
Trichloroisocyanuric acid, TCICA, also known as “swimming pool chlorine,” is a stable solid that oxidizes alcohols, following a mechanism similar to oxidation by HOCl. No reaction occurs between TCICA and the alcohol (in a solvent such as acetonitrile) until one drop of HCl(aq) is added, whereupon the reaction is over in a few minutes. Write the mechanism for this oxidation that shows the key role of the acid catalyst. Show the oxidation of just one alcohol, not three. (Hint: The carbonyls are the most basic sites on TCICA.)
Show how you would convert (S)-heptan-2-ol to
(a) (S)-2-chloroheptane.
(b) (R)-2-bromoheptane.
(c) (R)-heptan-2-ol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.