Chapter 11: Q27P (page 570)
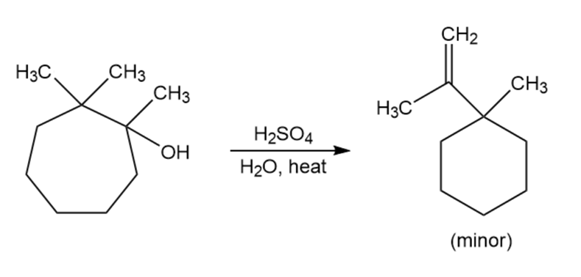
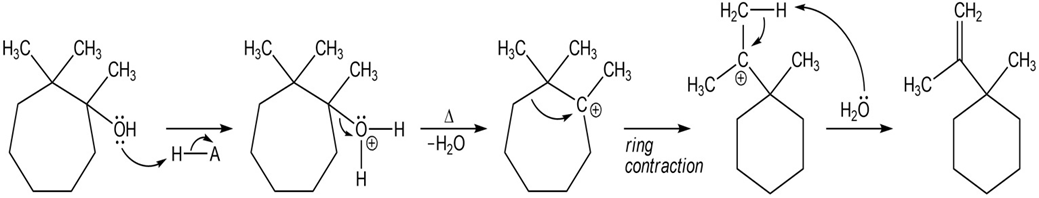
When the following substituted cycloheptanol undergoes dehydration, one of the minor products has undergone a ring contraction. Propose a mechanism to show how this ring contraction occurs.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q27P (page 570)
When the following substituted cycloheptanol undergoes dehydration, one of the minor products has undergone a ring contraction. Propose a mechanism to show how this ring contraction occurs.


All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would synthesize the following compounds. As starting materials, you may use any alcohols containing four or fewer carbon atoms, cyclohexanol, and any necessary solvents and inorganic reagents.
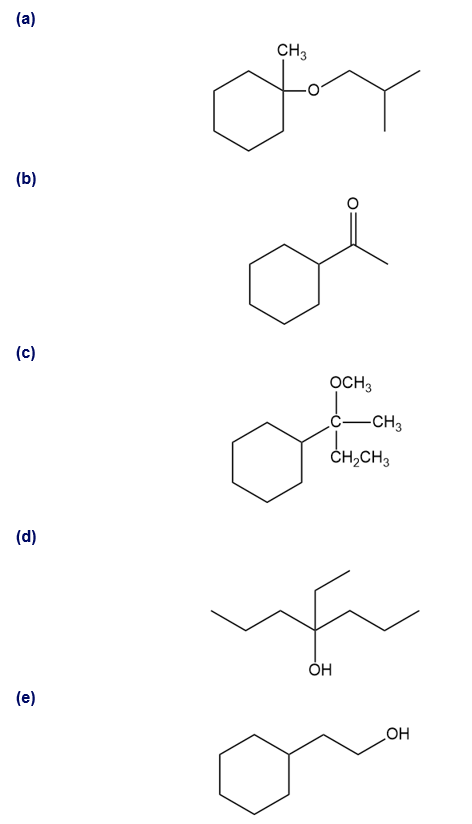
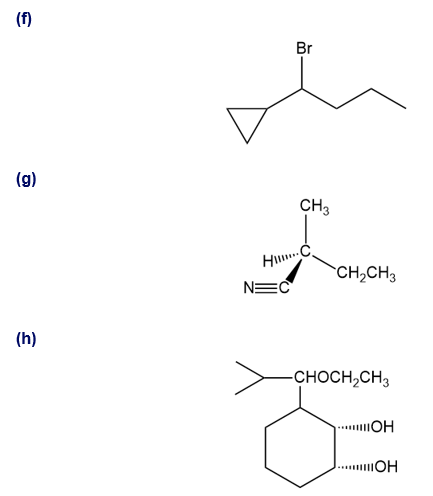
(a)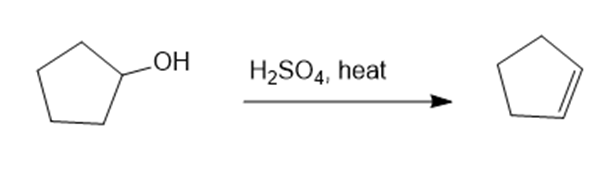
(b)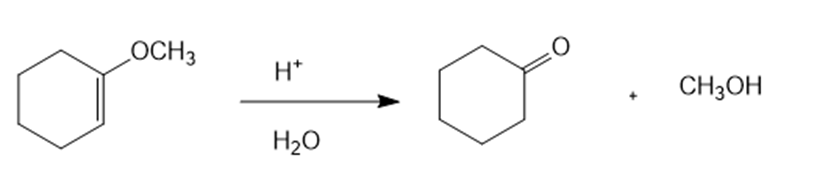
(c)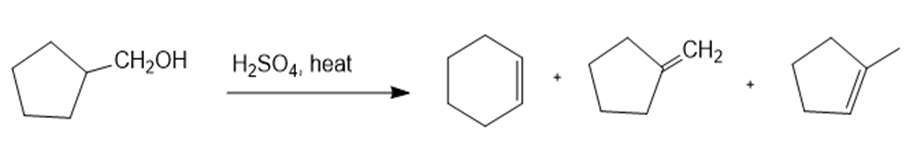
(d) 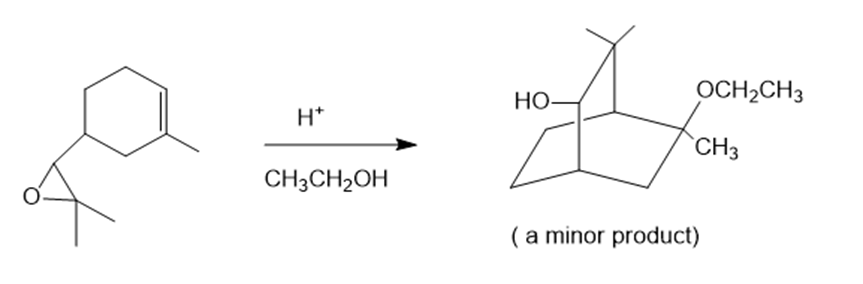
Neopentyl alcohol, (CH3)3CCH2OH, reacts with concentrated HBr to give 2-bromo-2-methylbutane, a rearranged product. Propose a mechanism for the formation of this product.
Predict the products of the sulfuric acid-catalyzed dehydration of the following alcohols. When more than one product is expected, label the major and minor products.
(a) 2-methylbutan-2-ol (b) pentan-1-ol (c) pentan-2-ol
(d) 1-isopropylcyclohexanol (e) 2-methylcyclohexanol
To practice working through the early parts of a multistep synthesis, devise syntheses of
(a) pentan-3-one from alcohols containing no more than three carbon atoms.
(b) 3-ethylpentan-2-one from compounds containing no more than three carbon atoms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.