Chapter 11: Q28P (page 571)
Propose a mechanism for each reaction.
(a) 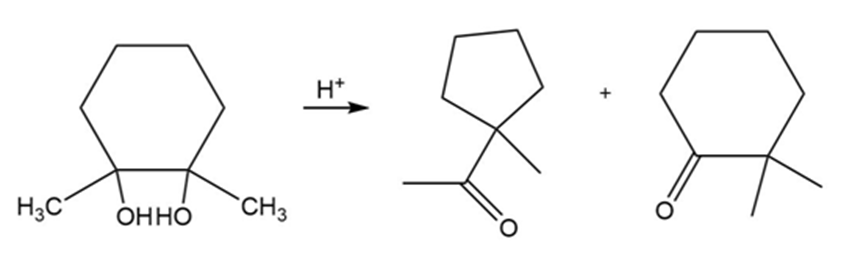
(b) 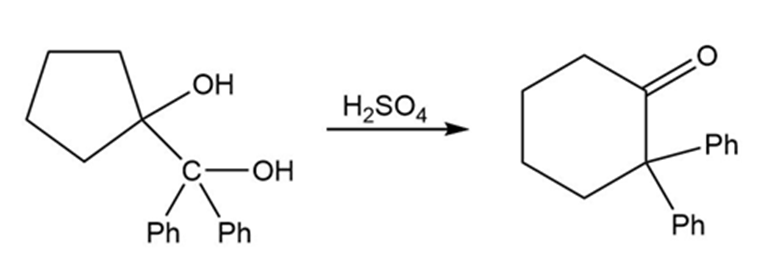
Short Answer
(a)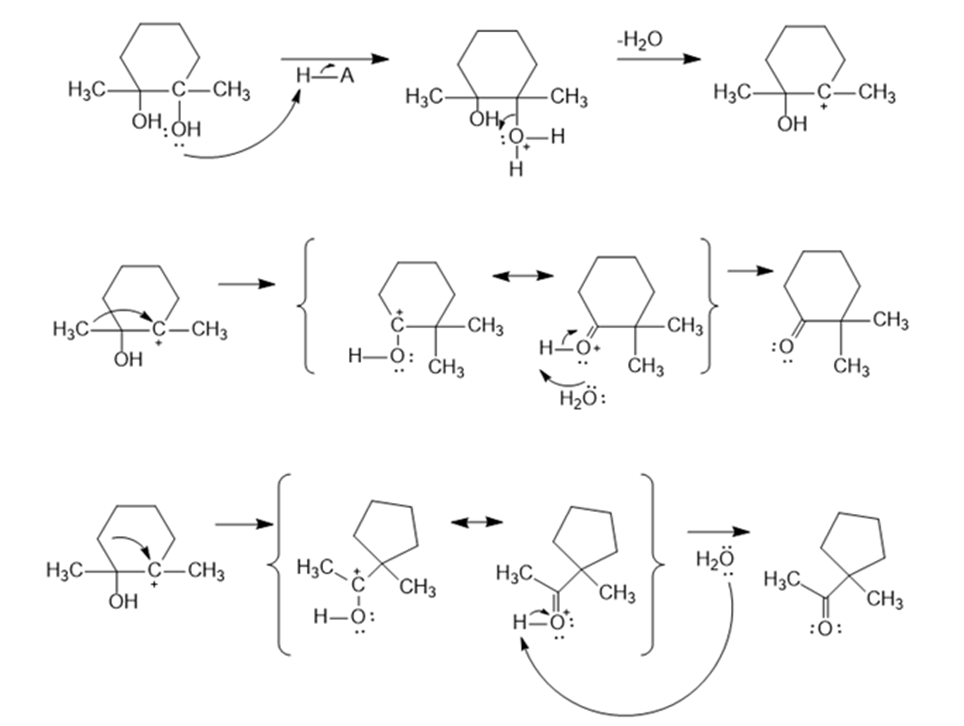
(b)
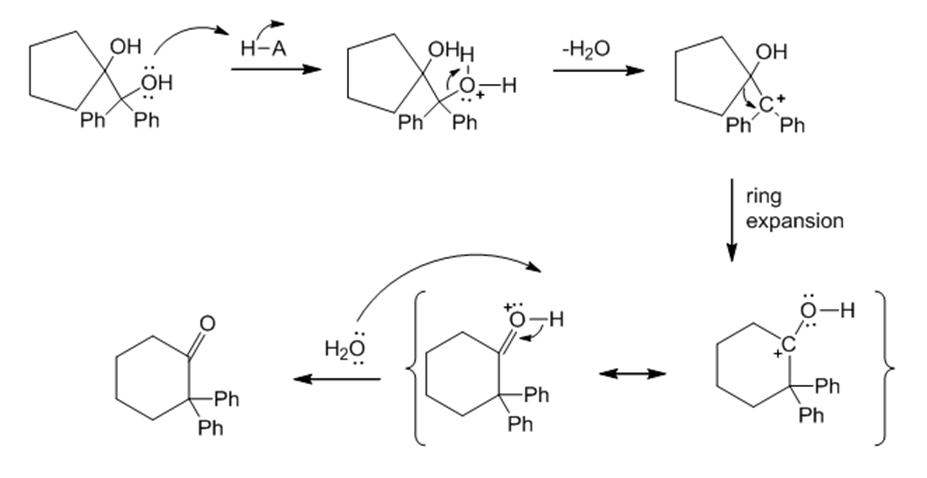
Learning Materials
Features
Discover
Chapter 11: Q28P (page 571)
Propose a mechanism for each reaction.
(a) 
(b) 
(a)
(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Write balanced equations for the three preceding reactions.
A chronic alcoholic requires a much larger dose of ethanol as an antidote to methanol poisoning than does a nonalcoholic patient. Suggest a reason why a larger dose of the competitive inhibitor is required for an alcoholic.
When 1-cyclohexylethanol is treated with concentrated aqueous, the major product is 1-bromo-1-ethylcyclohexane.
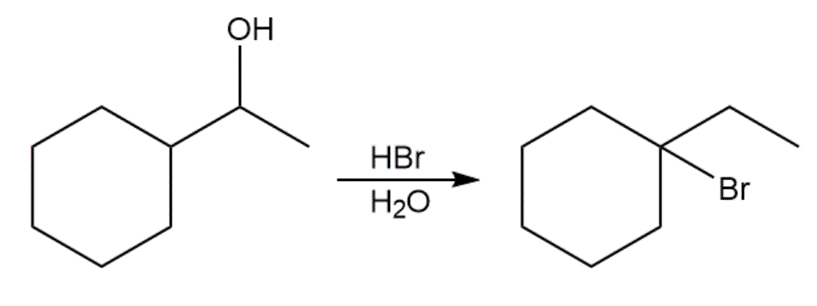
(a)Propose a mechanism for this reaction.
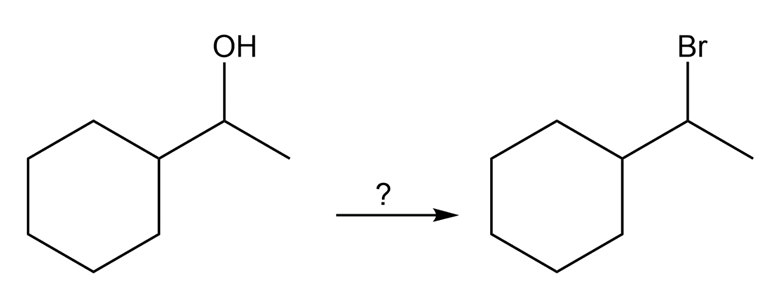
(b) How would you convert 1-cyclohexylethanol to (1-bromoethyl)cyclohexane in good yield?
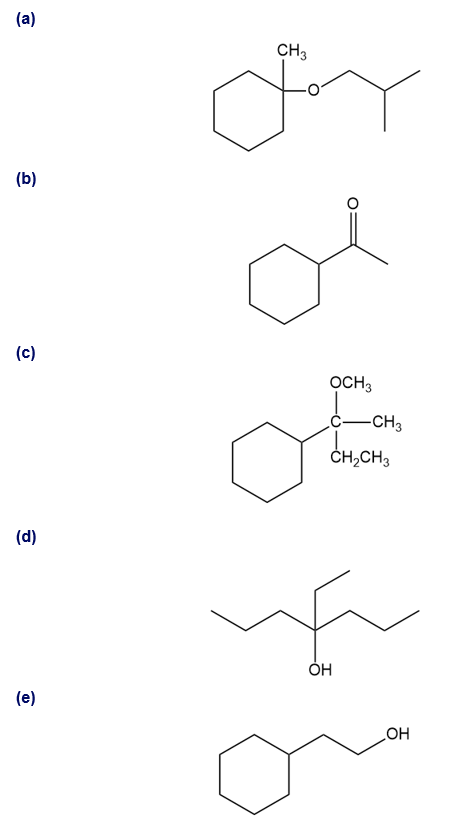
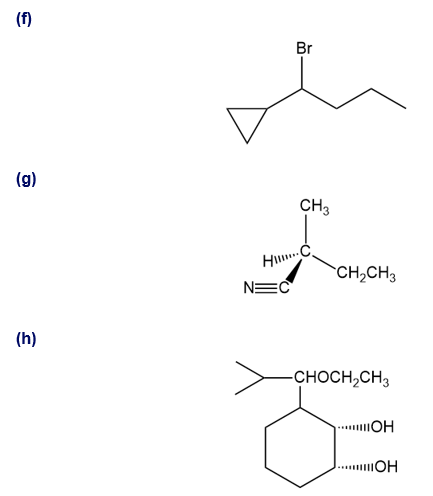
Show how you would synthesize the following compounds. As starting materials, you may use any alcohols containing four or fewer carbon atoms, cyclohexanol, and any necessary solvents and inorganic reagents.
Neopentyl alcohol, (CH3)3CCH2OH, reacts with concentrated HBr to give 2-bromo-2-methylbutane, a rearranged product. Propose a mechanism for the formation of this product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.