Chapter 11: Q15P (page 559)
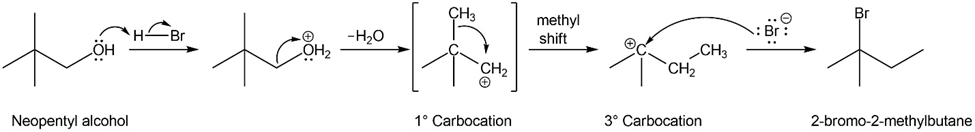
Neopentyl alcohol, (CH3)3CCH2OH, reacts with concentrated HBr to give 2-bromo-2-methylbutane, a rearranged product. Propose a mechanism for the formation of this product.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q15P (page 559)
Neopentyl alcohol, (CH3)3CCH2OH, reacts with concentrated HBr to give 2-bromo-2-methylbutane, a rearranged product. Propose a mechanism for the formation of this product.

All the tools & learning materials you need for study success - in one app.
Get started for free
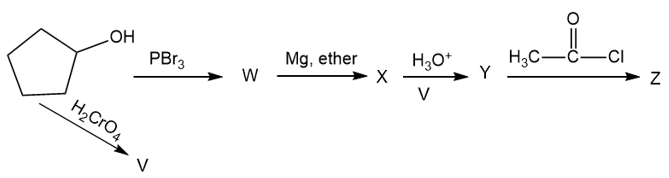
Give the structures of the intermediates and products V through Z.
(a) The reaction of butan-2-ol with concentrated aqueous HBr goes with partial racemization, giving more inversion than retention of configuration. Propose a mechanism that accounts for racemization with excess inversion.
(b)Under the same conditions, an optically active sample of trans-2-bromocyclopentanol reacts with concentrated aqueous HBr to give an optically inactive product, (racemic) trans-1,2-dibromocyclopentane. Propose a mechanism to show how this reaction goes with apparently complete retention of configuration, yet with racemization. (Hint: Draw out the mechanism of the reaction of cyclopentene with in water to give the starting material, trans-2- bromocyclopentanol. Consider how parts of this mechanism might be involved in the reaction with HBr.)
Propose a mechanism for each reaction.
(a) 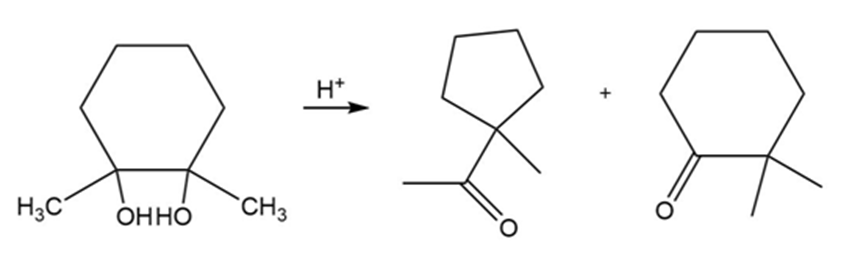
(b) 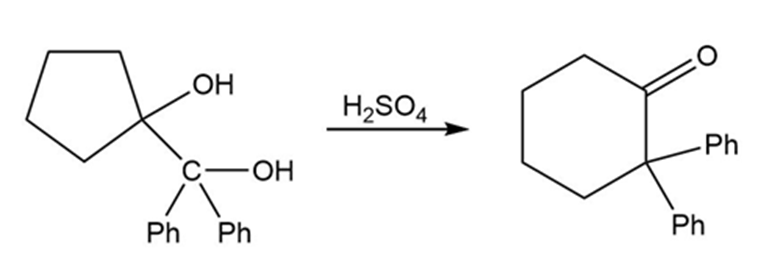
Predict the products formed by periodic acid cleavage of the following diols
(a) 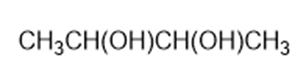
(b) 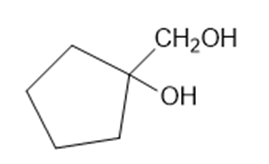
(c) 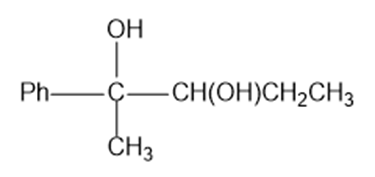
(d) 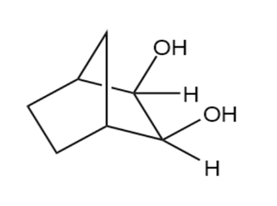
Write balanced equations for the three preceding reactions.
What do you think about this solution?
We value your feedback to improve our textbook solutions.