Chapter 11: Q16P (page 559)
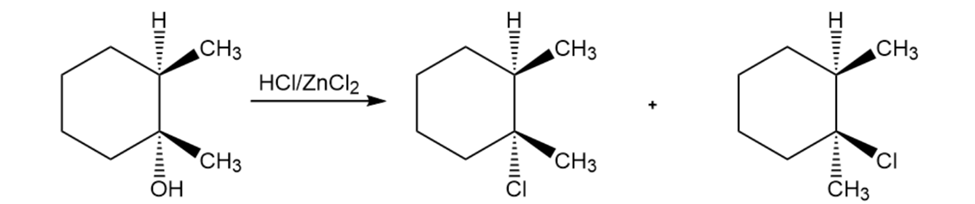
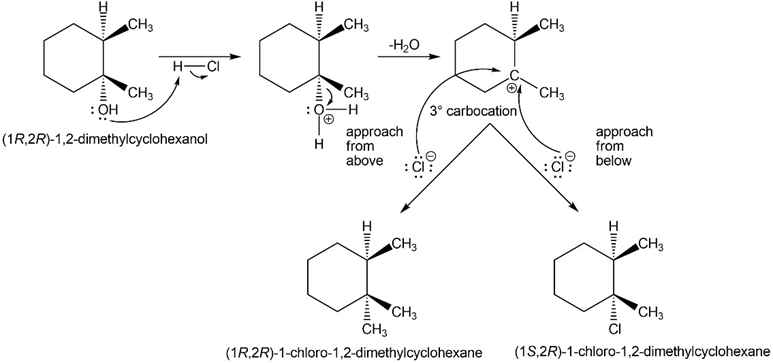
Explain the products observed in the following reaction of an alcohol with the Lucas reagent.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q16P (page 559)
Explain the products observed in the following reaction of an alcohol with the Lucas reagent.


All the tools & learning materials you need for study success - in one app.
Get started for free
The Williamson ether synthesis involves the displacement of an alkyl halide or tosylate by an alkoxide ion. Would the synthesis shown be possible by making a tosylate and displacing it? If so, show the sequence of reactions. If not, explain why not and show an alternative synthesis that would be more likely to work.

Predict the products of the sulfuric acid-catalyzed dehydration of the following alcohols. When more than one product is expected, label the major and minor products.
(a) 2-methylbutan-2-ol (b) pentan-1-ol (c) pentan-2-ol
(d) 1-isopropylcyclohexanol (e) 2-methylcyclohexanol
Predict the major products of dehydration catalyzed by sulfuric acid.
(a) heptan-1-ol
(b) heptan-2-ol
(c) pentan-2-ol
(d) 1-methylcyclohexanol
(e) Cyclohexylmethanol
(f) 2-methylcyclohexanol
A chronic alcoholic requires a much larger dose of ethanol as an antidote to methanol poisoning than does a nonalcoholic patient. Suggest a reason why a larger dose of the competitive inhibitor is required for an alcoholic.
Propose a mechanism for the reaction of
(a) 1-methyl cyclohexanol with HBr to form 1- bromo-1-methylcyclohexane.
(b) 2- cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.