Chapter 11: Q17P (page 559)
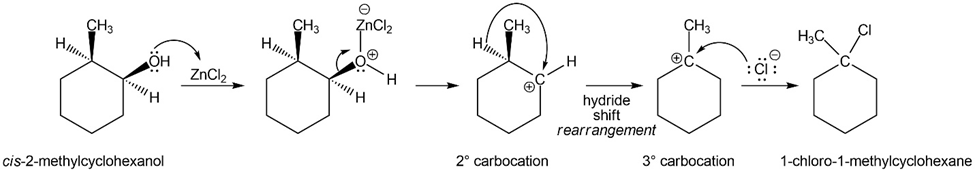
When cis-2-methylcyclohexanol reacts with the Lucas reagent, the major product is 1-chloro-1-methylcyclohexane. Propose a mechanism to explain the formation of this product.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q17P (page 559)
When cis-2-methylcyclohexanol reacts with the Lucas reagent, the major product is 1-chloro-1-methylcyclohexane. Propose a mechanism to explain the formation of this product.

All the tools & learning materials you need for study success - in one app.
Get started for free
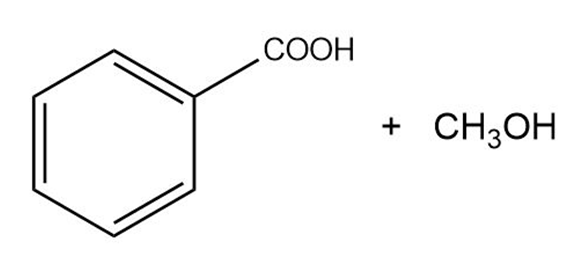
Predict the esterification products of the following acid/alcohol pairs.
(a) CH3CH2COOH + CH3OH
(b) CH3CH2OH + HNO3
(c) 2CH3CH2CH2OH + H3PO4
(d)
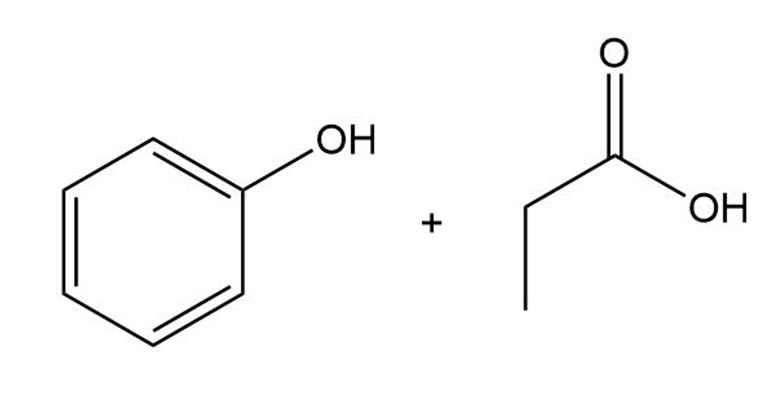
(e)
Unlike ethylene glycol, propylene glycol (propane-1, 2-diol) is non toxic because it oxidizes to a common metabolic intermediate. Give the structures of the biological oxidation products of propylene glycol.
Give the structures of the products you would expect when each alcohol reacts with
(1) HCl, ZnCl2; (2) HBr; (3) PBr3; (4) P/I2; and (5) SOCl2.
(a) butan-1-ol (b) 2-methylbutan-2-ol
(c) 2,2-dimethylbutan-1-ol (d) cis-3-methylcyclopentanol
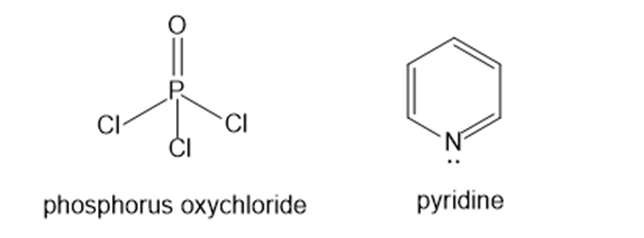
Some alcohols undergo rearrangement or other unwanted side reactions when they dehydrate in acid. Alcohols may be dehydrated under mildly basic conditions using phosphorus oxy-chloride (POCl3) in pyridine. The alcohol reacts with phosphorus oxychlorides much like it reacts with tosyl chloride (Section 11-5), displacing a chloride ion from phosphorus to give an alkyl dichlorophosphate ester. The dichlorophosphate group is an outstanding leaving group. Pyridine reacts as a base with the dichlorophosphate ester to give an E2 elimination. Propose a mechanism for the dehydration of cyclohexanol by POCl3 in pyridine.
Explain the products observed in the following reaction of an alcohol with the Lucas reagent.

What do you think about this solution?
We value your feedback to improve our textbook solutions.