Chapter 11: Q8P (page 551)
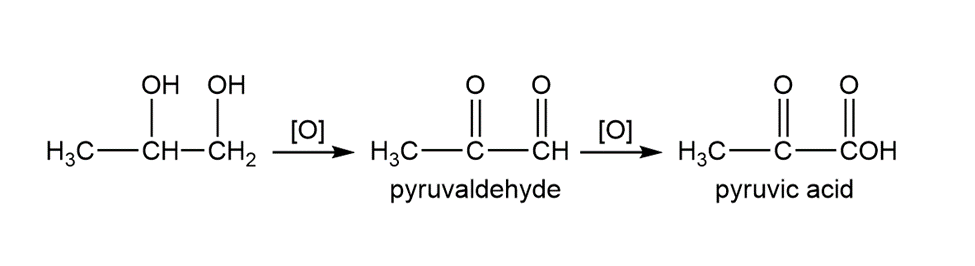
Unlike ethylene glycol, propylene glycol (propane-1, 2-diol) is non toxic because it oxidizes to a common metabolic intermediate. Give the structures of the biological oxidation products of propylene glycol.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q8P (page 551)
Unlike ethylene glycol, propylene glycol (propane-1, 2-diol) is non toxic because it oxidizes to a common metabolic intermediate. Give the structures of the biological oxidation products of propylene glycol.

All the tools & learning materials you need for study success - in one app.
Get started for free
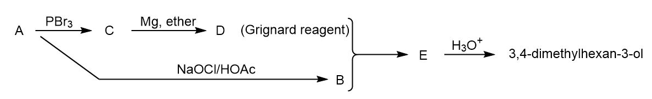
Compound A is an optically active alcohol. Treatment with chromic acid converts A into a ketone, B. In a separate reaction, A is treated with PBr3, converting A into compound C. Compound C is purified, and then it is allowed to react with magnesium in ether to give a Grignard reagent, D. Compound B is added to the resulting solution of the Grignard reagent. After hydrolysis of the initial product (E), this solution is found to contain 3,4 dimethylhexan-3-ol. Propose structures for compounds A, B, C, D, and E.
Use resonance forms of the conjugate bases to explain why methanesulfonic acid (CH3SO3H,pKa= -2.6) is a much stronger acid than acetic acid (CH3COOH,pKa =4.8)
Show the alcohol and the acid chloride that combine to make the following esters.
(a) 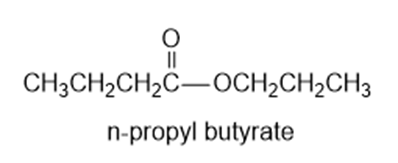
(b) 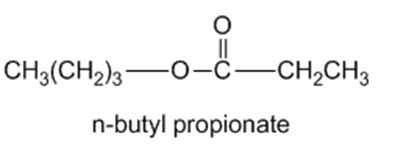
(c) 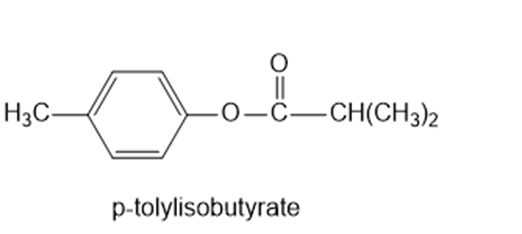
(d) 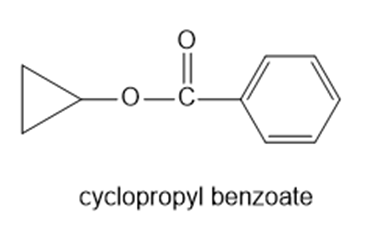
Predict the products of the following reactions.
(a) cyclohexylmethanol + TsCl / pyridine (b) product of (a) + LiAlH4
(c) 1- methylcyclohexanol + H2SO4, heat (d) product of (c) + H2, Pt
When 1-cyclohexylethanol is treated with concentrated aqueous, the major product is 1-bromo-1-ethylcyclohexane.
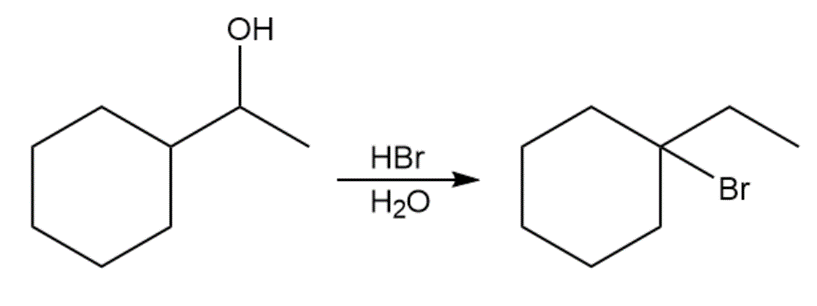
(a)Propose a mechanism for this reaction.
(b) How would you convert 1-cyclohexylethanol to (1-bromoethyl)cyclohexane in good yield?
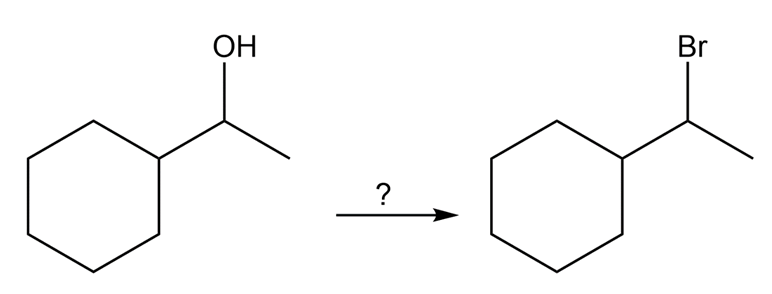
What do you think about this solution?
We value your feedback to improve our textbook solutions.