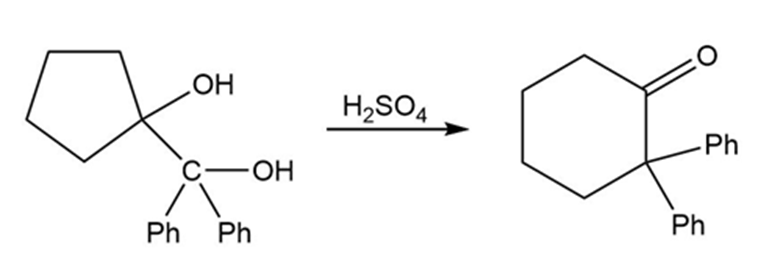
Chapter 11: Q52P (page 589)
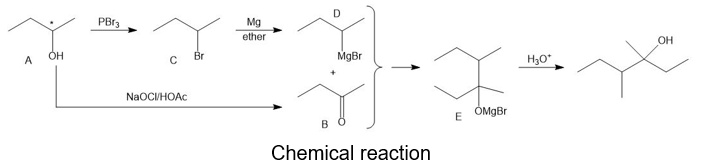
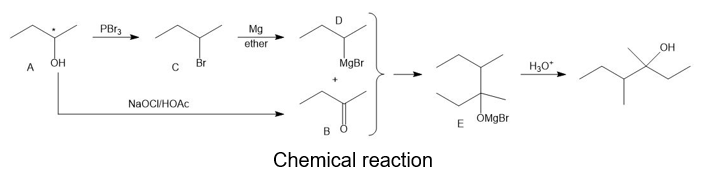
Compound A is an optically active alcohol. Treatment with chromic acid converts A into a ketone, B. In a separate reaction, A is treated with PBr3, converting A into compound C. Compound C is purified, and then it is allowed to react with magnesium in ether to give a Grignard reagent, D. Compound B is added to the resulting solution of the Grignard reagent. After hydrolysis of the initial product (E), this solution is found to contain 3,4 dimethylhexan-3-ol. Propose structures for compounds A, B, C, D, and E.
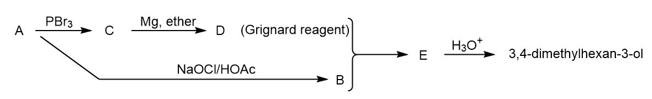
Short Answer