Chapter 11: Q51P (page 589)
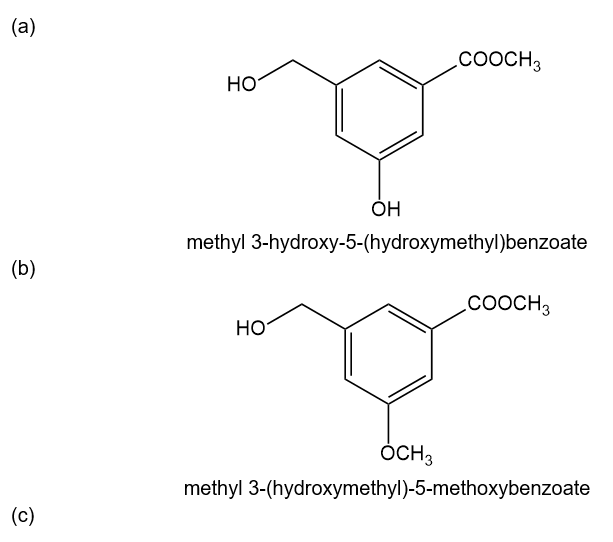
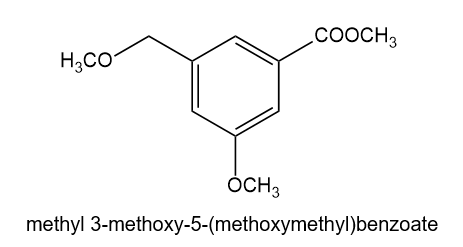
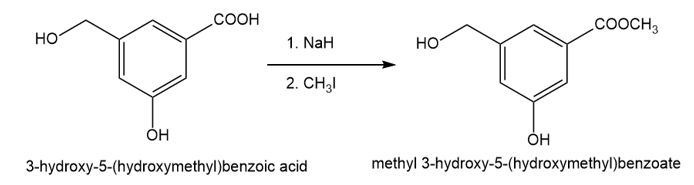
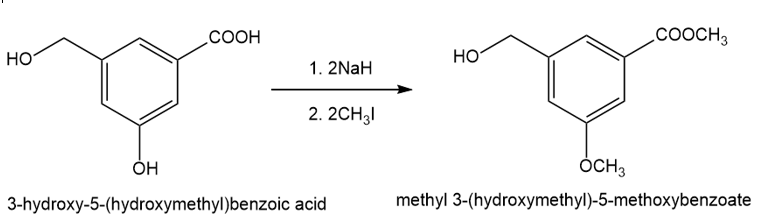
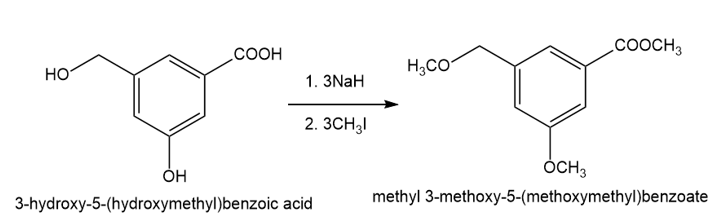
The compound shown below has three different types of OH groups, all with different acidities. Show the structure produced after this compound is treated with different amounts of NaH followed by a methylating reagent. Add a brief explanation.
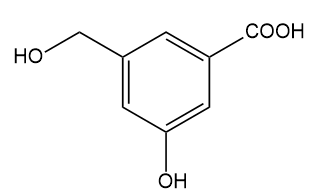
(a)1 equivalent of NaH, followed by 1 equivalent of CH3l and heat
(b)2 equivalents of NaH, followed by 2 equivalents of CH3l and heat
(c) 3 equivalents of NaH, followed by 3 equivalents of CH3l and heat
Short Answer