Chapter 11: Q57P (page 590)
Show how you would synthesize the following compound. As starting materials, you may use any alcohols containing five or fewer carbon atoms and any necessary solvents and inorganic reagents.
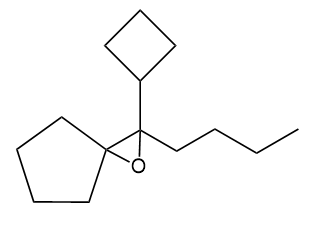
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q57P (page 590)
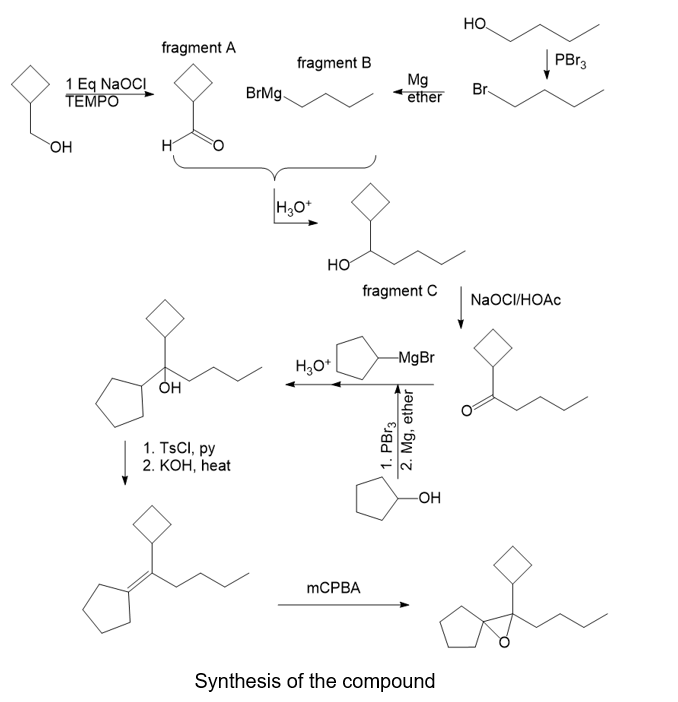
Show how you would synthesize the following compound. As starting materials, you may use any alcohols containing five or fewer carbon atoms and any necessary solvents and inorganic reagents.


All the tools & learning materials you need for study success - in one app.
Get started for free
Compound A is an optically active alcohol. Treatment with chromic acid converts A into a ketone, B. In a separate reaction, A is treated with PBr3, converting A into compound C. Compound C is purified, and then it is allowed to react with magnesium in ether to give a Grignard reagent, D. Compound B is added to the resulting solution of the Grignard reagent. After hydrolysis of the initial product (E), this solution is found to contain 3,4 dimethylhexan-3-ol. Propose structures for compounds A, B, C, D, and E.
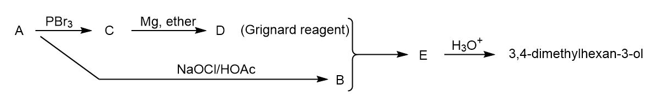
Predict the products you expect when the following starting material undergoes oxidation with an excess of each of the reagents shown below.
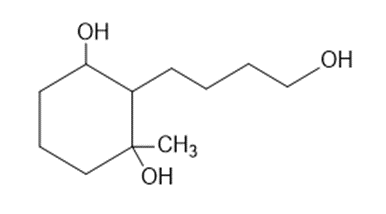
(a)chromic acid
(b)PCC(pyridinium chlorochromate)
(c)sodium hypochlorite/ acetic acid
(d)DMSO and oxalyl chloride
(e)DMP(periodinane)reagent
Under normal circumstances, tertiary alcohols are not oxidized. However, when the tertiary alcohol is allylic, it can undergo a migration of the double bond (called an allylic shift) and subsequent oxidation of the alcohol. A particularly effective reagent for this reaction is Bobbitt’s reagent, similar to TEMPO used in many oxidations. (M. Shibuya et al., J. Org. Chem., 2008, 73, 4750.)
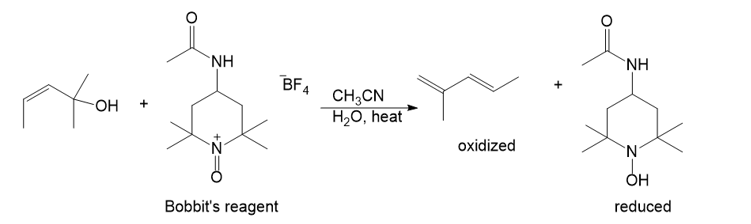
Show the expected product when each of these 3° allylic alcohols is oxidized by Bobbitt’s reagent.
(a)
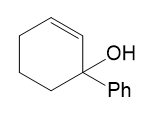
(b)
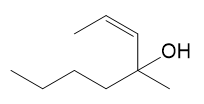
(c)
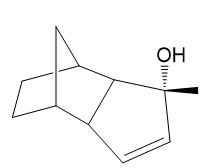
(d)
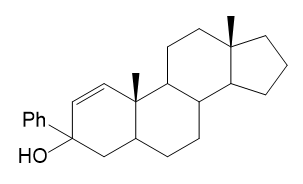
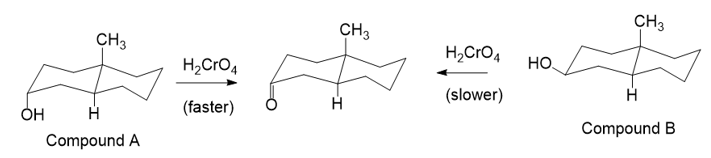
Chromic acid oxidation of alcohol (Section 11-2A) occurs in two steps: formation of the chromate ester, followed by an elimination of chromium. Which step do you expect to be rate-limiting? Careful kinetic studies have shown that Compound A undergoes chromic acid oxidation over 10 times as fast as Compound B. Explain this large difference in rates.
Use resonance forms of the conjugate bases to explain why methanesulfonic acid (CH3SO3H,pKa= -2.6) is a much stronger acid than acetic acid (CH3COOH,pKa =4.8)
What do you think about this solution?
We value your feedback to improve our textbook solutions.