The acidic strength decides the stability of a conjugate base. The acid comprising a more stable conjugate base is highly stable.
The methanesulfonate ion is stabilized by resonance and induction. It has three resonance structures. The sulfur atom is more electronegative than carbon and comprises a small role in stabilizing the negative charge on carbon.
The acetate ion has two resonance structures. It does not have an inductive effect to stabilize the anion. The resonance structures of acetate ion and methylsulfonate ion can be given as:
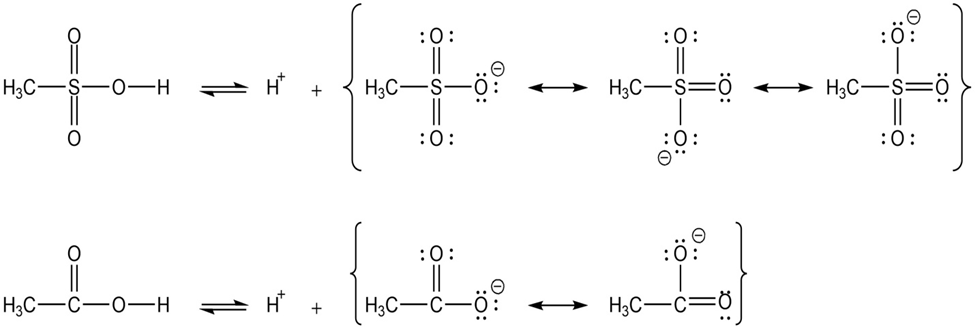
Resonance structures of acetate and methylsulfonate ion
Acetic acid is a mild acid, but methanesulfonic acid is stronger than acetic acid.



