Chapter 11: Q31P (page 573)
Show the alcohol and the acid chloride that combine to make the following esters.
(a) 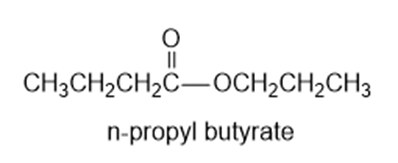
(b) 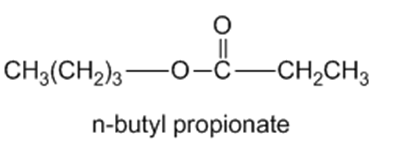
(c) 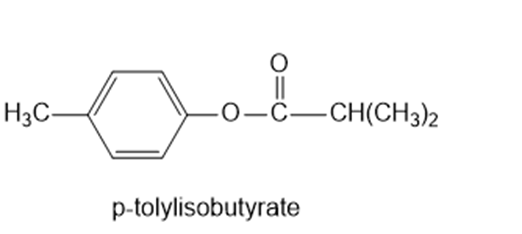
(d) 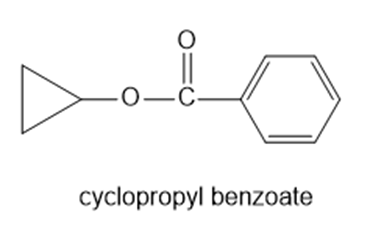
Short Answer
(a) 
(b) 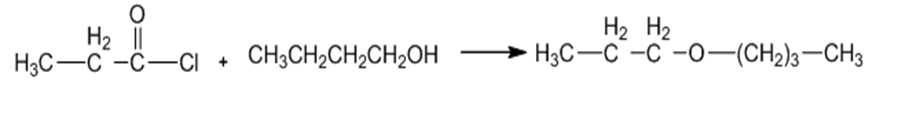
(c) 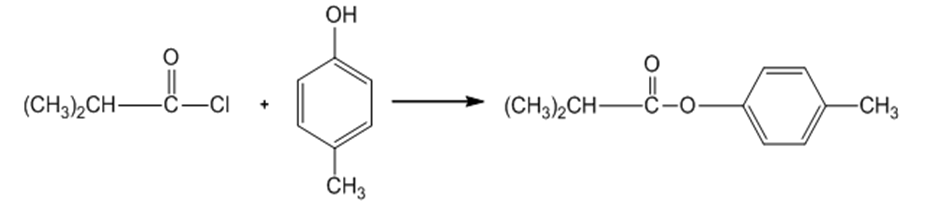
(d) 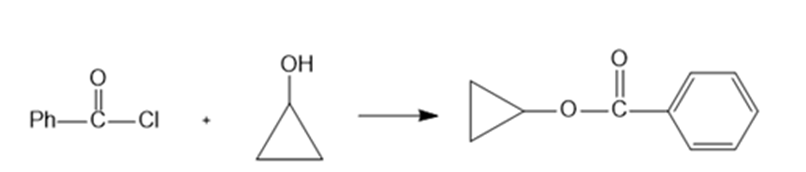
Learning Materials
Features
Discover
Chapter 11: Q31P (page 573)
Show the alcohol and the acid chloride that combine to make the following esters.
(a) 
(b) 
(c) 
(d) 
(a) 
(b) 
(c) 
(d) 
All the tools & learning materials you need for study success - in one app.
Get started for free
The following pseudo-syntheses (guaranteed not to work) exemplify a common conceptual error.
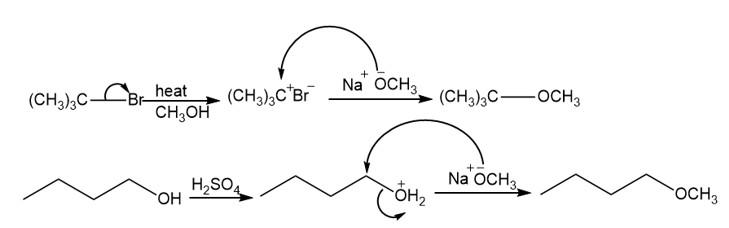
(a)What is the conceptual error implicit in these syntheses?
(b)Propose syntheses that are more likely to succeed.
Alcohols combine with ketones and aldehydes to form interesting derivatives, which we will discuss in Chapter 18. The following reactions show the hydrolysis of two such derivatives. Propose mechanisms for these reactions.
(a)
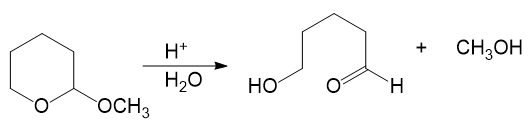
(b)
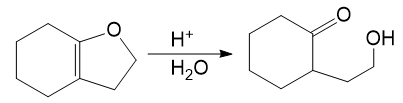
Suggest the most appropriate method for each of the following laboratory syntheses. In each case, suggest both a chromium reagent and a chromium-free reagent
(a) butan - 1 - ol → butanal, CH3CH2CH2CHO
(b) but - 2 - en - 1 - ol → but - 2 - enoic acid, CH3CH = CHCOOH
(c) butan - 2 - ol → butan - 2 - one, CH3COCH2CH3
(d) cyclopentanol → 1 - cyclopentylpropan - 1 - ol (twosteps)
(e) cyclopentyclohexanol → 2 - methylcyclohexanone (several steps)
(f) 1 - methylcyclohexanol → 2 - methylcyclohexanone (several steps)
Predict the products you expect when the following starting material undergoes oxidation with an excess of each of the reagents shown below.
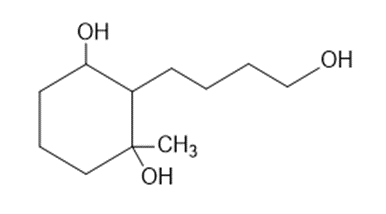
(a)chromic acid
(b)PCC(pyridinium chlorochromate)
(c)sodium hypochlorite/ acetic acid
(d)DMSO and oxalyl chloride
(e)DMP(periodinane)reagent
Contrast the mechanism of the two preceding reactions, the dehydration and condensation of ethanol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.