Chapter 11: Q24P (page 567)
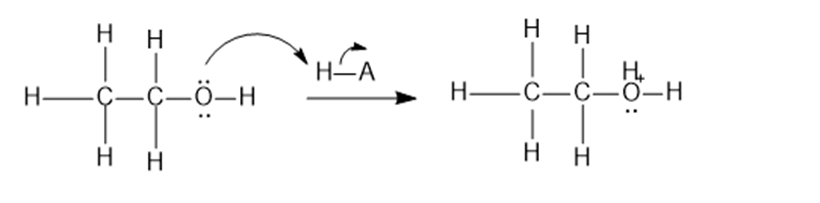
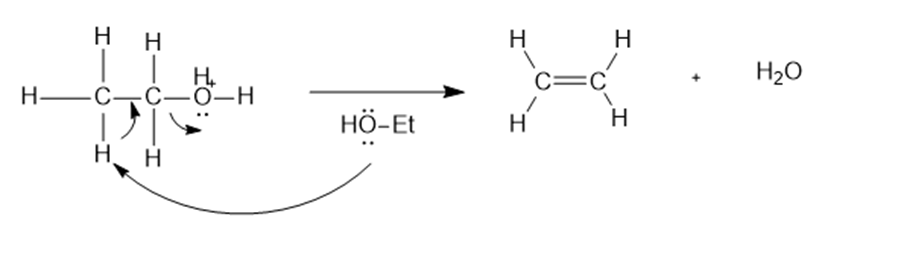
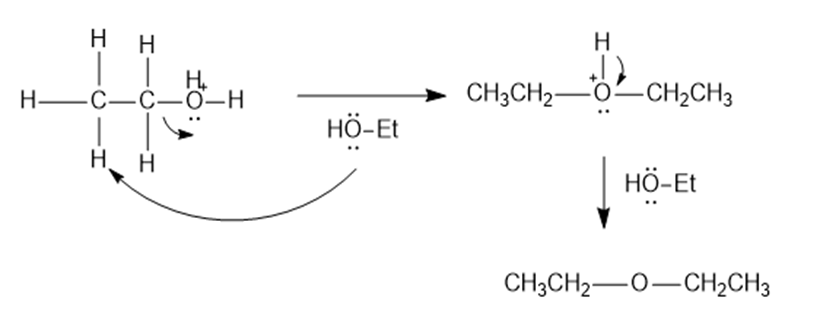
Contrast the mechanism of the two preceding reactions, the dehydration and condensation of ethanol.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q24P (page 567)
Contrast the mechanism of the two preceding reactions, the dehydration and condensation of ethanol.



All the tools & learning materials you need for study success - in one app.
Get started for free
Use resonance forms of the conjugate bases to explain why methanesulfonic acid (CH3SO3H,pKa= -2.6) is a much stronger acid than acetic acid (CH3COOH,pKa =4.8)
Propose a mechanism for the reaction of
(a) 1-methyl cyclohexanol with HBr to form 1- bromo-1-methylcyclohexane.
(b) 2- cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane.
Predict the products formed by periodic acid cleavage of the following diols
(a) 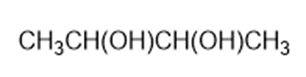
(b) 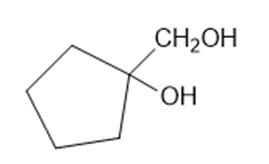
(c) 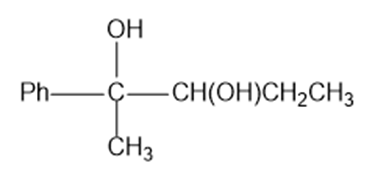
(d) 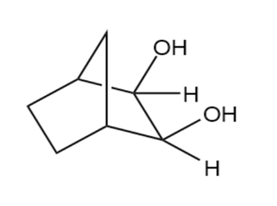
Predict the products you expect when the following starting material undergoes oxidation with an excess of each of the reagents shown below.
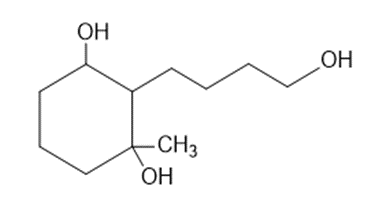
(a)chromic acid
(b)PCC(pyridinium chlorochromate)
(c)sodium hypochlorite/ acetic acid
(d)DMSO and oxalyl chloride
(e)DMP(periodinane)reagent
Give the structures of the products you would expect when each alcohol reacts with
(1) HCl, ZnCl2; (2) HBr; (3) PBr3; (4) P/I2; and (5) SOCl2.
(a) butan-1-ol (b) 2-methylbutan-2-ol
(c) 2,2-dimethylbutan-1-ol (d) cis-3-methylcyclopentanol
What do you think about this solution?
We value your feedback to improve our textbook solutions.