Chapter 11: Q54P (page 589)
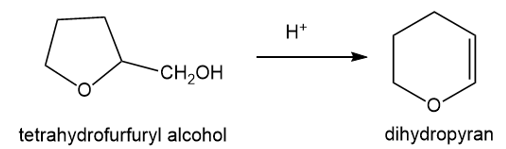
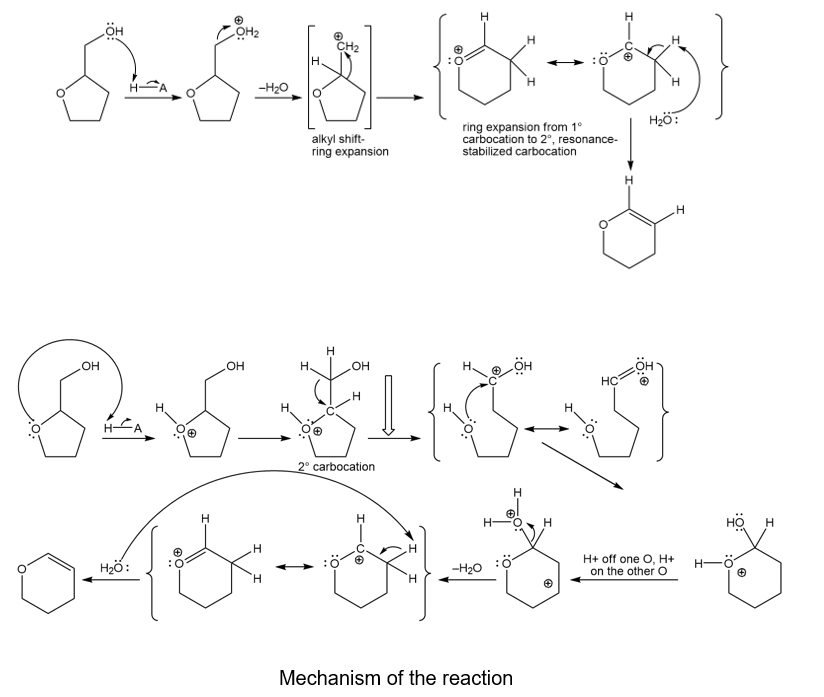
Under acid catalysis, tetrahydrofurfuryl alcohol reacts to give surprisingly good yields of dihydropyran. Propose a mechanism to explain this useful synthesis.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q54P (page 589)
Under acid catalysis, tetrahydrofurfuryl alcohol reacts to give surprisingly good yields of dihydropyran. Propose a mechanism to explain this useful synthesis.


All the tools & learning materials you need for study success - in one app.
Get started for free
Give the structure of the principal product(s) when each of the following alcohols reacts with (1) Na2Cr2O7/H2SO4 (2) PCC, (3) DMP, and (4) 1 equiv NaOCl-TEMPO.
(a)octan-1-ol (b)octan-3-ol
(c) 4-hydroxydecanal (d) 1-methylcyclohexan-1,4-diol
To practice working through the early parts of a multistep synthesis, devise syntheses of
(a) pentan-3-one from alcohols containing no more than three carbon atoms.
(b) 3-ethylpentan-2-one from compounds containing no more than three carbon atoms.
Suggest the most appropriate method for each of the following laboratory syntheses. In each case, suggest both a chromium reagent and a chromium-free reagent
(a) butan - 1 - ol → butanal, CH3CH2CH2CHO
(b) but - 2 - en - 1 - ol → but - 2 - enoic acid, CH3CH = CHCOOH
(c) butan - 2 - ol → butan - 2 - one, CH3COCH2CH3
(d) cyclopentanol → 1 - cyclopentylpropan - 1 - ol (twosteps)
(e) cyclopentyclohexanol → 2 - methylcyclohexanone (several steps)
(f) 1 - methylcyclohexanol → 2 - methylcyclohexanone (several steps)
Predict the products of the following reactions.
(a) cyclohexylmethanol + TsCl / pyridine (b) product of (a) + LiAlH4
(c) 1- methylcyclohexanol + H2SO4, heat (d) product of (c) + H2, Pt
Two products are observed in the following reaction.
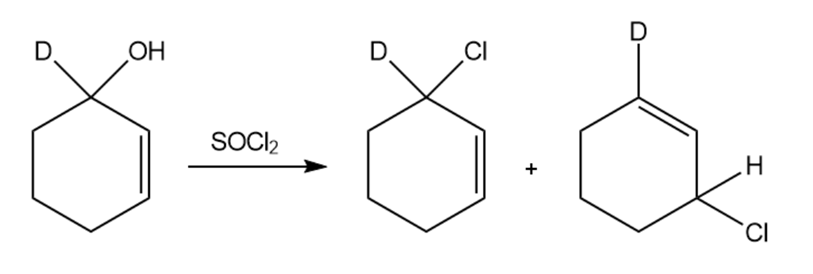
(a) Suggest a mechanism to explain how these two products are formed.
(b) Your mechanism for part (a) should be different from the usual mechanism of the reaction of SOCl2 with alcohols. Explain why the reaction follows a different mechanism in
this case.
What do you think about this solution?
We value your feedback to improve our textbook solutions.